How many sigma and pi bonds are in \[Se{O_3}^{2 - }\]?
Answer
579.9k+ views
Hint: We need to understand what are sigma and pi bonds and how do we calculate them. In addition, to study about $\left( \sigma \right)$ and $\left( \Pi \right)$ bonds we need to understand molecular orbitals and molecular orbital theory. Understanding of atomic orbitals is also necessary. The first covalent bond between two atoms is always a sigma bond $\left( \sigma \right)$ . Any second or third bond is known as a pi bond. Hence, we would first need to understand the bonding in the given molecule and accordingly calculate the number of sigma and pi bonds in \[Se{O_3}^{2 - }\].
Complete step by step answer:
The first covalent bond between two atoms is always a sigma bond $\left( \sigma \right)$. Any second or third bond is known as a pi bond. Let us explain this with the help of a simple $HCN$ molecule. This molecule has one single and one triple bond given as follows $H - C \equiv N$ . As we know that the first covalent bond is always a sigma bond, the single bond between $H$ and $C$ is definitely a sigma bond and the first bond between $C$ and $N$ is a sigma bond and the remaining two are pi bonds.
The given molecule is a selenite ion \[Se{O_3}^{2 - }\] . Its skeletal structure is given below:
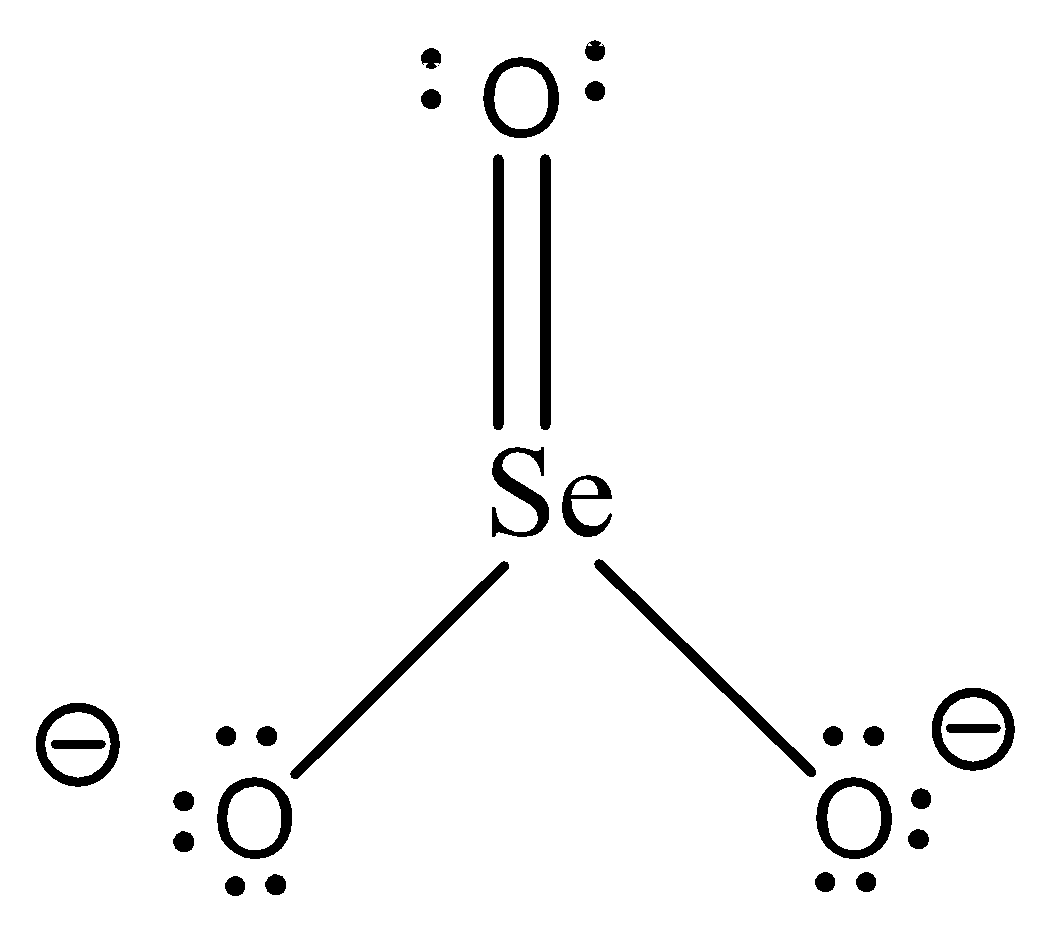
As seen in the skeletal structure, there are 2 single bonds and one double bond. Hence the 2 single bonds are sigma bonds along with the first bond of the double bond and the second bond of the double bond is a pi bond.
Hence, there are three sigma bonds and one pi bond in \[Se{O_3}^{2 - }\] .
Note: It must be noted that there is a simple trick to calculate the number of sigma bonds in a molecule. The formula for this trick is \[S = x - 1\] where $S - $ the number of sigma bonds and $x$ is the number of atoms in the molecule. The number of molecules \[Se{O_3}^{2 - }\] is $4$. According to the formula, $x = 4$ .Therefore, $S = 4 - 1 = 3$ sigma bonds and one pi bond.
Complete step by step answer:
The first covalent bond between two atoms is always a sigma bond $\left( \sigma \right)$. Any second or third bond is known as a pi bond. Let us explain this with the help of a simple $HCN$ molecule. This molecule has one single and one triple bond given as follows $H - C \equiv N$ . As we know that the first covalent bond is always a sigma bond, the single bond between $H$ and $C$ is definitely a sigma bond and the first bond between $C$ and $N$ is a sigma bond and the remaining two are pi bonds.
The given molecule is a selenite ion \[Se{O_3}^{2 - }\] . Its skeletal structure is given below:

As seen in the skeletal structure, there are 2 single bonds and one double bond. Hence the 2 single bonds are sigma bonds along with the first bond of the double bond and the second bond of the double bond is a pi bond.
Hence, there are three sigma bonds and one pi bond in \[Se{O_3}^{2 - }\] .
Note: It must be noted that there is a simple trick to calculate the number of sigma bonds in a molecule. The formula for this trick is \[S = x - 1\] where $S - $ the number of sigma bonds and $x$ is the number of atoms in the molecule. The number of molecules \[Se{O_3}^{2 - }\] is $4$. According to the formula, $x = 4$ .Therefore, $S = 4 - 1 = 3$ sigma bonds and one pi bond.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE




