What is the shape of the $Cl{{F}_ {3}} $ molecule?
(A) trigonal planar
(B) Trigonal pyramidal
(C) T-shaped
(D) Tetrahedral
Answer
590.4k+ views
Hint: To solve this question refer to the valence shell electron pair repulsion theory or VSEPR theory as it is used to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms.
Complete answer:
We have been provided with $Cl{{F}_ {3}} $ molecule,
We need to tell the shape of this molecule,
So, for that:
Firstly, we will be using the VSEPR theory,
We know that VSEPR theory predict the geometry of an individual molecule from the number of electron pairs surrounding the central atoms,
So, we have the molecule of $Cl{{F}_ {3}} $,
So, according to the VSEPR theory,
The electron groups in $Cl{{F}_ {3}} $ are 5 around the central Cl atom,
Out of which 3 are bonding molecule and 2 are non-bonding molecule,
Also, there are 2 lone pair presents in the $Cl{{F}_ {3}} $ molecule,
These lone pair are adjusted on the axial plane to maximize the bond angle and minimize the bond repulsion,
Out of 3 fluorine atoms, two fluorine atoms are adjusted on equatorial position separated by angle of ${{180} ^ {\circ}} $,
And the third fluorine atom is adjusted on axial plane in a see-saw manner,
It would acquire a trigonal bipyramidal with 5 electron groups around the Cl but due to the presence of lone pairs its shape is T-shaped.
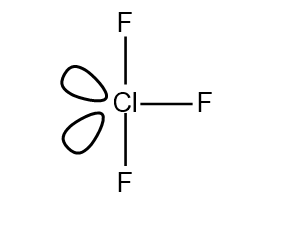
So, we can say that the shape of the $Cl{{F}_ {3}} $ molecule is t-shaped .
So, the correct option is (C).
Note:
VSEPR theory have certain limitations too:
(1) this theory was unable to explain about the elements that have the same number of electrons.
(2) this theory does not explain the transition metal compounds.
Complete answer:
We have been provided with $Cl{{F}_ {3}} $ molecule,
We need to tell the shape of this molecule,
So, for that:
Firstly, we will be using the VSEPR theory,
We know that VSEPR theory predict the geometry of an individual molecule from the number of electron pairs surrounding the central atoms,
So, we have the molecule of $Cl{{F}_ {3}} $,
So, according to the VSEPR theory,
The electron groups in $Cl{{F}_ {3}} $ are 5 around the central Cl atom,
Out of which 3 are bonding molecule and 2 are non-bonding molecule,
Also, there are 2 lone pair presents in the $Cl{{F}_ {3}} $ molecule,
These lone pair are adjusted on the axial plane to maximize the bond angle and minimize the bond repulsion,
Out of 3 fluorine atoms, two fluorine atoms are adjusted on equatorial position separated by angle of ${{180} ^ {\circ}} $,
And the third fluorine atom is adjusted on axial plane in a see-saw manner,
It would acquire a trigonal bipyramidal with 5 electron groups around the Cl but due to the presence of lone pairs its shape is T-shaped.

So, we can say that the shape of the $Cl{{F}_ {3}} $ molecule is t-shaped .
So, the correct option is (C).
Note:
VSEPR theory have certain limitations too:
(1) this theory was unable to explain about the elements that have the same number of electrons.
(2) this theory does not explain the transition metal compounds.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




