How many right angles are there in a \[Xe{F_5}^ + \] ion?
Answer
507.3k+ views
Hint: Bases on the valence electrons of all atoms in a molecule, the molecular geometry and electron geometry will be expected. \[Xe{F_5}^ + \] has octahedral as electron geometry and square pyramidal as molecular geometry. Based on the molecular geometry, right angles can be calculated.
Complete answer:
\[Xe{F_5}^ + \] is a molecule consisting of Xenon as a central metal atom, and five fluorine atoms. The valence electrons on Xenon are \[8\] , and five fluorine atoms have \[5 \times 7 = 35\] electrons, as each fluorine atom has \[7\] valence electrons. Thus, the total valence electrons on the given molecule will be \[8 + 35 - 1 = 42\] electrons.
The eight electrons on a xenon atom are involved in bond formation with five fluorine atoms and one electron was lost as it is a cation, the remaining two electrons will exist as a lone pair of electrons. Thus, the hybridization is \[s{p^3}{d^2}\] . Thus, the electron geometry is octahedral, and the molecular geometry is square pyramidal due to the presence of one lone pair of electrons.
The structure of \[Xe{F_5}^ + \] is
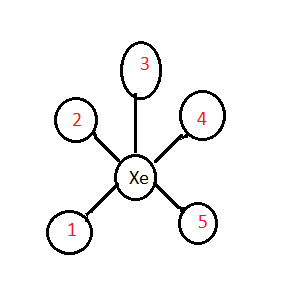
The five fluorine atoms were considered as \[1,2,3,4,\] and \[5\] . Based on the above structure the right angles were:
\[3 - Xe - 1\]
\[3 - Xe - 2\]
\[3 - Xe - 4\]
\[3 - Xe - 5\]
\[1 - Xe - 2\]
\[1 - Xe - 5\]
\[4 - Xe - 5\]
\[2 - Xe - 4\]
Thus, there is a total of eight right angles in \[Xe{F_5}^ + \]
Note:
Right angle means the angle must be equal to \[{90^0}\] . In the above structure, there were eight right angles, meaning there were eight bonds in which the bond angle equal to \[{90^0}\] . The right angles can be considered based on the molecular geometry but not electron geometry.
Complete answer:
\[Xe{F_5}^ + \] is a molecule consisting of Xenon as a central metal atom, and five fluorine atoms. The valence electrons on Xenon are \[8\] , and five fluorine atoms have \[5 \times 7 = 35\] electrons, as each fluorine atom has \[7\] valence electrons. Thus, the total valence electrons on the given molecule will be \[8 + 35 - 1 = 42\] electrons.
The eight electrons on a xenon atom are involved in bond formation with five fluorine atoms and one electron was lost as it is a cation, the remaining two electrons will exist as a lone pair of electrons. Thus, the hybridization is \[s{p^3}{d^2}\] . Thus, the electron geometry is octahedral, and the molecular geometry is square pyramidal due to the presence of one lone pair of electrons.
The structure of \[Xe{F_5}^ + \] is

The five fluorine atoms were considered as \[1,2,3,4,\] and \[5\] . Based on the above structure the right angles were:
\[3 - Xe - 1\]
\[3 - Xe - 2\]
\[3 - Xe - 4\]
\[3 - Xe - 5\]
\[1 - Xe - 2\]
\[1 - Xe - 5\]
\[4 - Xe - 5\]
\[2 - Xe - 4\]
Thus, there is a total of eight right angles in \[Xe{F_5}^ + \]
Note:
Right angle means the angle must be equal to \[{90^0}\] . In the above structure, there were eight right angles, meaning there were eight bonds in which the bond angle equal to \[{90^0}\] . The right angles can be considered based on the molecular geometry but not electron geometry.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




