How many resonating structures are possible for the compound.

Answer
564k+ views
Hint: To solve this question, we first need to know what are resonating structures. Resonating structures or resonance structures can be used to depict the delocalization of electrons through a set of Lewis structures in an ion or a molecule containing multiple atoms.
Complete answer:
Sometimes, when a single Lewis structure cannot depict how atoms are bonded in an ion or a molecule due to the presence of fractional bonds or partial charges, the chemical bonding is depicted by resonance structure.
When the resonance structures of a molecule or an ion are merged to form a resonance hybrid.
A lone pair, when added to an atom, creates a negative charge on the molecule. Similarly, when a lone pair is removed from an atom, a positive charge is created on the atom.
When a bond is broken, a lone pair of electrons are added to the atom. Similarly, when a lone pair is added to an atom, a bond is formed.
Bonds and lone pairs delocalized in a molecule to form resonating structures. A resonance cycle is said to be complete when the original molecule is formed and the bonds and the lone pairs are the same as the original molecule.
It is important to note that while counting the resonating structures, the final structure which is the same as the original structure of the molecule should not be counted as one of the resonating structures.
The resonating structures of furan are as follows:
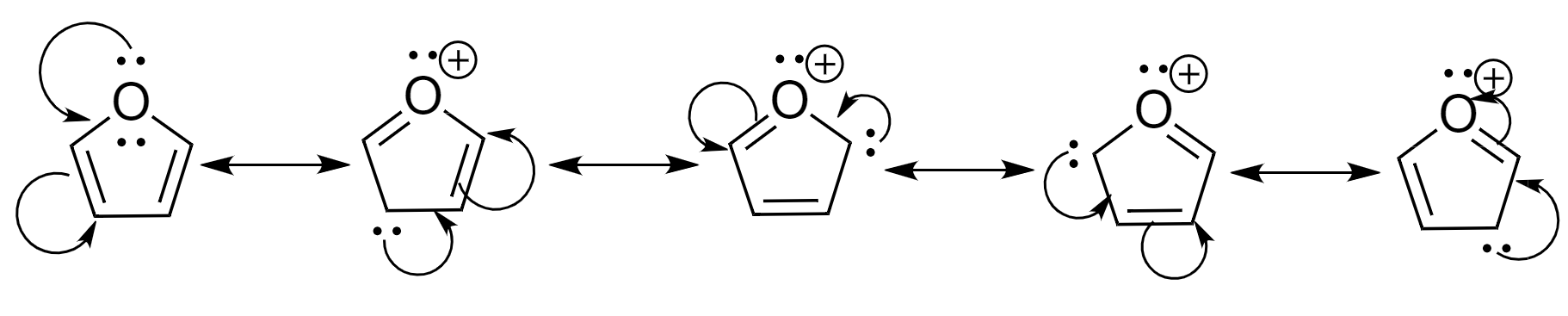
Hence, we can see that furan forms 5 resonating structures.
Note:
Some of the common mistakes that should be kept in mind while drawing the resonating structure of a molecule are
1. Atoms should not be moved around while drawing the resonating structure.
2. The charge and the number of lone pairs on the molecule must not change.
3. The curved arrows should be drawn correctly.
Complete answer:
Sometimes, when a single Lewis structure cannot depict how atoms are bonded in an ion or a molecule due to the presence of fractional bonds or partial charges, the chemical bonding is depicted by resonance structure.
When the resonance structures of a molecule or an ion are merged to form a resonance hybrid.
A lone pair, when added to an atom, creates a negative charge on the molecule. Similarly, when a lone pair is removed from an atom, a positive charge is created on the atom.
When a bond is broken, a lone pair of electrons are added to the atom. Similarly, when a lone pair is added to an atom, a bond is formed.
Bonds and lone pairs delocalized in a molecule to form resonating structures. A resonance cycle is said to be complete when the original molecule is formed and the bonds and the lone pairs are the same as the original molecule.
It is important to note that while counting the resonating structures, the final structure which is the same as the original structure of the molecule should not be counted as one of the resonating structures.
The resonating structures of furan are as follows:

Hence, we can see that furan forms 5 resonating structures.
Note:
Some of the common mistakes that should be kept in mind while drawing the resonating structure of a molecule are
1. Atoms should not be moved around while drawing the resonating structure.
2. The charge and the number of lone pairs on the molecule must not change.
3. The curved arrows should be drawn correctly.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




