How many resonance structures exist for the formate ion, \[HC{{O}_{2}}^{-}\]?
Answer
594.3k+ views
Hint Formate is the anion i.e. negatively charged ion derived from formic acid. The IUPAC name of formate ion is methanoate and represented as\[HCO{{O}^{-}}\], \[CHO{{O}^{-}}\] and \[HC{{O}_{2}}^{-}\]. It is the product of deprotonation of formic acid.
Complete Step by step solution: Resonance structures are sets of Lewis structures which describe the delocalization of electrons in polyatomic ions or molecules. In many cases a single Lewis structure fails to explain the bonding in a molecule due to presence of partial charge and fractional bonds in it. In these cases resonance structure helps to describe the chemical bonding in that molecule.
The formate ion is composed of one atom of hydrogen and carbon and two atoms of oxygen in which the ion contains a charge of -1. It can be able to form resonance structures due to the presence of a lone pair in oxygen that can relocate or delocalize as a double bond between itself and the carbon atom which will cause the movement of the pi bond to move to the other oxygen atom as a lone pair. There are two resonance structure of formate ion are possible which can be shown as:
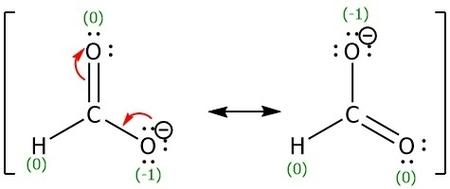
Negative charge is placed on the most electronegative element i.e. on oxygen.
Note: Resonance describes the bonding in particular molecules or ions by merging a number of contributory structures or forms which are jointly known as canonical structures or resonance structures within the theory of valence bonding into a hybrid resonance also known as hybrid structure.
Complete Step by step solution: Resonance structures are sets of Lewis structures which describe the delocalization of electrons in polyatomic ions or molecules. In many cases a single Lewis structure fails to explain the bonding in a molecule due to presence of partial charge and fractional bonds in it. In these cases resonance structure helps to describe the chemical bonding in that molecule.
The formate ion is composed of one atom of hydrogen and carbon and two atoms of oxygen in which the ion contains a charge of -1. It can be able to form resonance structures due to the presence of a lone pair in oxygen that can relocate or delocalize as a double bond between itself and the carbon atom which will cause the movement of the pi bond to move to the other oxygen atom as a lone pair. There are two resonance structure of formate ion are possible which can be shown as:

Negative charge is placed on the most electronegative element i.e. on oxygen.
Note: Resonance describes the bonding in particular molecules or ions by merging a number of contributory structures or forms which are jointly known as canonical structures or resonance structures within the theory of valence bonding into a hybrid resonance also known as hybrid structure.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Explain zero factorial class 11 maths CBSE

What is the atomic mass of Sodium a 22 gmol b 23 gmol class 11 chemistry CBSE

What organs are located on the left side of your body class 11 biology CBSE

Bond order ofO2 O2+ O2 and O22 is in order A O2 langle class 11 chemistry CBSE

Draw the molecular orbital diagram of N2N2 + N2 Write class 11 chemistry CBSE




