Predict the product formed in the following reaction.
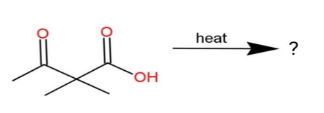
A. 3-methyl-2-butanone
B. 3, 3-dimethylmalonic acid
C. propane
D. 3, 3-dimethyl-2, 4-pentanedione

Answer
566.7k+ views
Hint: When an organic compound contains a ketonic group along with a carboxylic group, then it is called as a keto acid. The naming of keto acids can also be done as oxo alkanoic acids.
Complete step by step answer: We have been given a compound which is subjected to heat, and we have to find the product obtained. The compound contains a keto acid, with a 4 carbon chain along with 2 methyl branches. So, the name of this compound is 2,2-dimethyl-3-oxobutanoic acid. This keto acid when subjected to heat converts into di carboxylic acid, which is 3, 3-dimethylmalonic acid . The reaction is as,
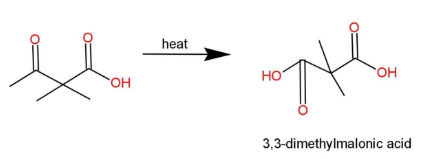
The reaction forms di carboxylic acid, as by heating of this keto acid, catalytic oxidation occurs, which creates an intermediate of carbanion, that has a hydroxyl group attached to form a stable 3, 3-dimethylmalonic acid.
Additional information: Keto acids are of various types, like alpha keto acids, that contain a keto group adjacent to the carboxyl group. Beta-keto acids that have a ketone group placed at the second carbon from the carboxyl group.
Hence, the reaction gives 3, 3-dimethylmalonic acid, so, option B is correct.
Note: Keto acids on heating have a removal of carboxyl ions. But here it is converted to 3, 3-dimethylmalonic acid, due to the compound being beta- keto acid heated at high temperature range produces an enol intermediate, hence the product here.
Complete step by step answer: We have been given a compound which is subjected to heat, and we have to find the product obtained. The compound contains a keto acid, with a 4 carbon chain along with 2 methyl branches. So, the name of this compound is 2,2-dimethyl-3-oxobutanoic acid. This keto acid when subjected to heat converts into di carboxylic acid, which is 3, 3-dimethylmalonic acid . The reaction is as,

The reaction forms di carboxylic acid, as by heating of this keto acid, catalytic oxidation occurs, which creates an intermediate of carbanion, that has a hydroxyl group attached to form a stable 3, 3-dimethylmalonic acid.
Additional information: Keto acids are of various types, like alpha keto acids, that contain a keto group adjacent to the carboxyl group. Beta-keto acids that have a ketone group placed at the second carbon from the carboxyl group.
Hence, the reaction gives 3, 3-dimethylmalonic acid, so, option B is correct.
Note: Keto acids on heating have a removal of carboxyl ions. But here it is converted to 3, 3-dimethylmalonic acid, due to the compound being beta- keto acid heated at high temperature range produces an enol intermediate, hence the product here.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




