Plot P versus $\dfrac{1}{V}$ at different temperatures.
Answer
587.4k+ views
Hint: We know Boyle’s Law:
According to Boyle's law, as the pressure decreases the volume increases thus, the bubbles at the bottom of a glass of a soft drink get larger when it rises to the surface because when the bubbles rise, the pressure inside the bubble starts to get reduced and the volume (size) of the bubble increases.
At constant temperature, the pressure and the volume of the gas are inversely related.
${\text{PV = K}}$
Where P is the pressure of the gas at a constant temperature.
V is the volume of the gas at a constant temperature.
k is the constant.
Complete step by step answer:
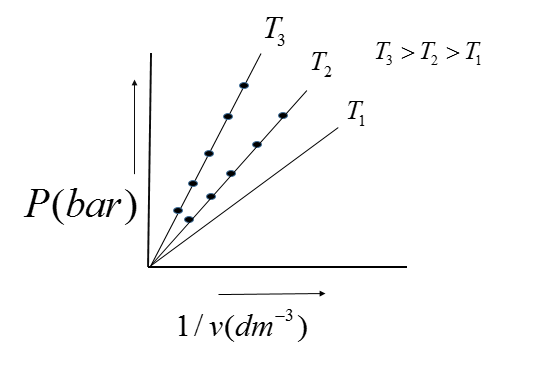
The graph is a straight line through the origin, verifying that the pressure is proportional to $\dfrac{1}{V}$ verifying Boyle's Law. Also, all values of pressure and volume are the same.
$P \propto$ $\dfrac{1}{V}$
$p = k\dfrac{1}{V}$
Therefore, when different values of pressure are plotted against the reciprocals of the respective $\dfrac{1}{V}$ volume. This graph justifies Boyle's law because the straight line is obtained which passes through the origin.
So, the correct answer is Option D.
Note: We need to know that the Boyles' law explains the reason why the bubble at the bottom of a glass of a soft drink gets larger when it rises to the surface and why the bag of potato chips on a plane ride appeared to be inflated when taken out, We need to know that the bag of potato chips on a plane ride appears to be inflated when taken out because according to Boyle's law whenever the pressure of a gaseous system decreases its volume increases at a constant temperature. At the higher height, the pressure is reduced so the gas inside the bag becomes inflated.
According to Boyle's law, as the pressure decreases the volume increases thus, the bubbles at the bottom of a glass of a soft drink get larger when it rises to the surface because when the bubbles rise, the pressure inside the bubble starts to get reduced and the volume (size) of the bubble increases.
At constant temperature, the pressure and the volume of the gas are inversely related.
${\text{PV = K}}$
Where P is the pressure of the gas at a constant temperature.
V is the volume of the gas at a constant temperature.
k is the constant.
Complete step by step answer:

The graph is a straight line through the origin, verifying that the pressure is proportional to $\dfrac{1}{V}$ verifying Boyle's Law. Also, all values of pressure and volume are the same.
$P \propto$ $\dfrac{1}{V}$
$p = k\dfrac{1}{V}$
Therefore, when different values of pressure are plotted against the reciprocals of the respective $\dfrac{1}{V}$ volume. This graph justifies Boyle's law because the straight line is obtained which passes through the origin.
So, the correct answer is Option D.
Note: We need to know that the Boyles' law explains the reason why the bubble at the bottom of a glass of a soft drink gets larger when it rises to the surface and why the bag of potato chips on a plane ride appeared to be inflated when taken out, We need to know that the bag of potato chips on a plane ride appears to be inflated when taken out because according to Boyle's law whenever the pressure of a gaseous system decreases its volume increases at a constant temperature. At the higher height, the pressure is reduced so the gas inside the bag becomes inflated.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE




