Phenyl isocyanide is prepared from aniline by:
(a)- Rosenmund’s reaction
(b)- Kolbe’s reaction
(c)- Reimer-Tiemann reaction
(d)- Fittig reaction
(e)- Carbylamine reaction.
Answer
631.5k+ views
Hint: The catalyst used is chloroform and an alcoholic solution of potassium hydroxide. This reaction is a type of test reaction that is used to distinguish between primary amines from secondary and tertiary amines.
Complete step by step solution:
Let us study all the options one by one:
(a)- Rosenmund reaction.
Acid chlorides are easily reduced to the corresponding aldehydes by passing hydrogen gas through boiling xylene solution of the acid chloride in the presence of a Pd catalyst supported over\[BaS{{O}_{4}}\].
\[RCOCl+{{H}_{2}}\to RCOH+HCl\]
(b)- Kolbe’s reaction.
Sodium phenoxide when heated with carbon dioxide at 400 K under a pressure of 4-7 atmosphere followed by acidification gives 2-hydroxybenzoic acid (salicylic acid) as the main product.
(c)- Reimer-Tiemann reaction.
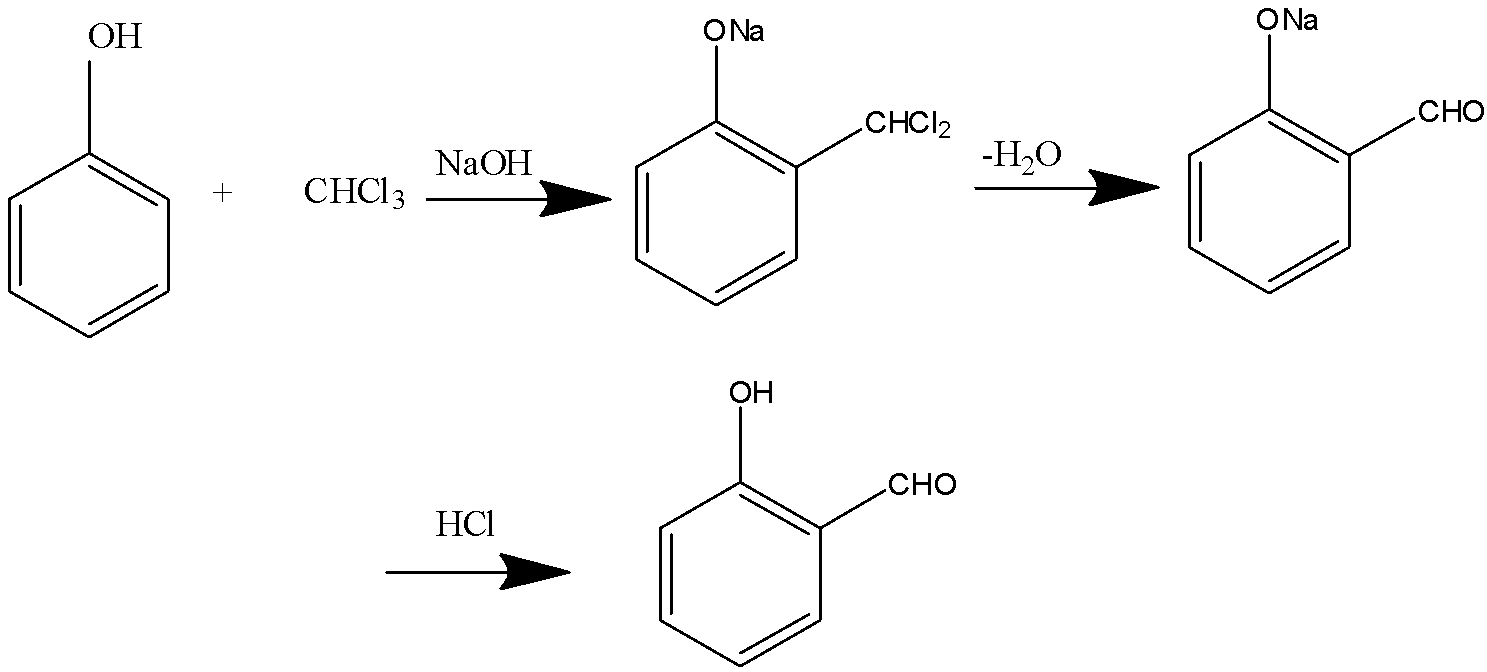
Treatment of phenol with chloroform in the presence of aqueous or potassium hydroxide at 340K followed by hydrolysis of the resulting product gives 2-hydroxybenzaldehyde (salicylaldehyde) as the major product.
(d)- Fittig reaction.
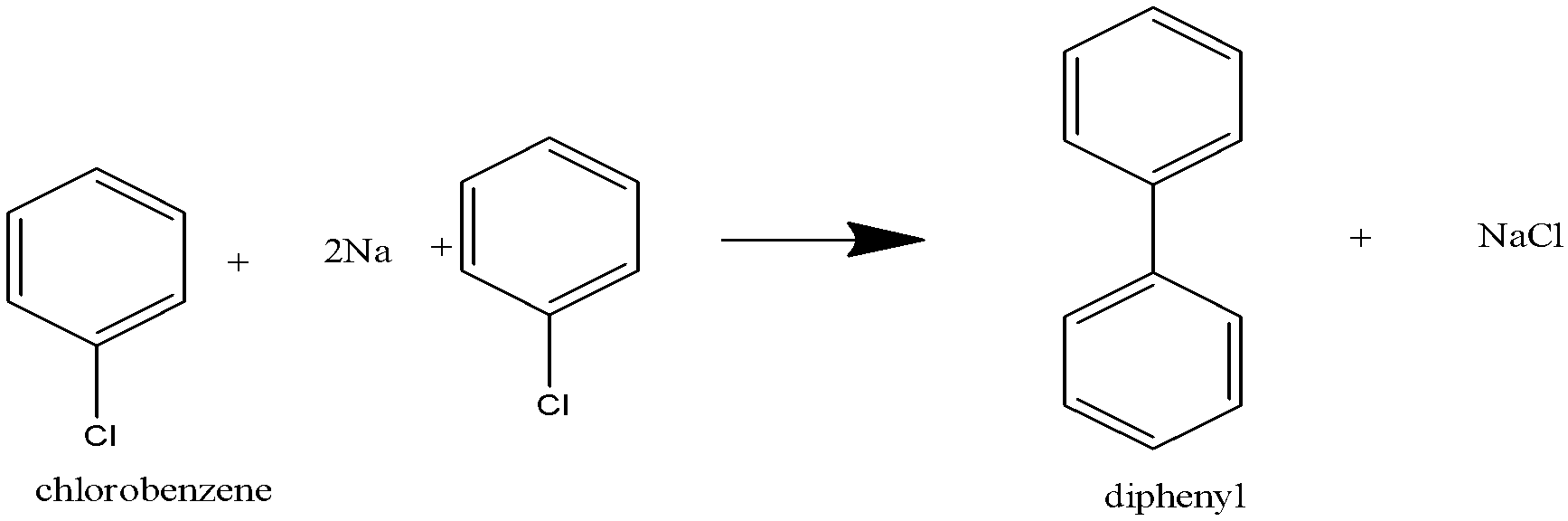
When only haloarenes are treated with sodium, diaryls are produced.
(e)- Carbylamine reaction.
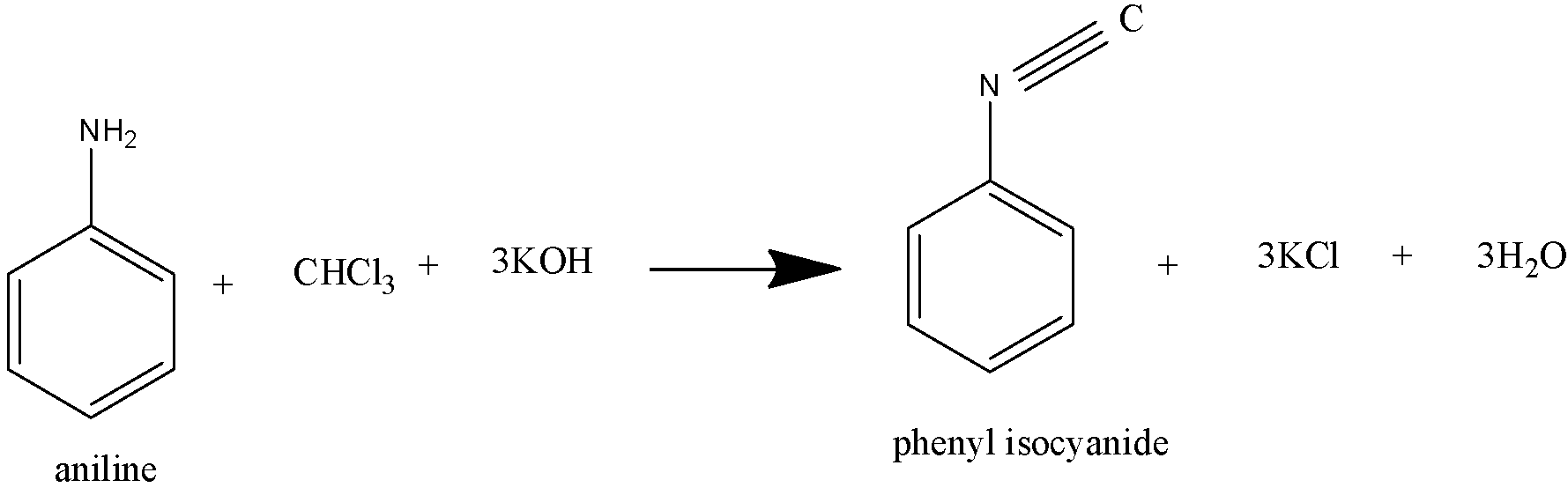
Both aliphatic and aromatic primary amines when warmed with chloroform and an alcoholic solution of KOH produce isocyanides or carbylamines.
So, from the above observation, we can see that aniline is converted into phenyl isocyanide in the carbylamine reaction.
So, the correct answer is “Option E”.
Note: Don’t get confused between reamer-teimann and carbylamines reaction because both of them have chloroform as one of the reactants. In contrast, secondary and tertiary amines do not give the carbylamine reaction.
Complete step by step solution:
Let us study all the options one by one:
(a)- Rosenmund reaction.
Acid chlorides are easily reduced to the corresponding aldehydes by passing hydrogen gas through boiling xylene solution of the acid chloride in the presence of a Pd catalyst supported over\[BaS{{O}_{4}}\].
\[RCOCl+{{H}_{2}}\to RCOH+HCl\]
(b)- Kolbe’s reaction.
Sodium phenoxide when heated with carbon dioxide at 400 K under a pressure of 4-7 atmosphere followed by acidification gives 2-hydroxybenzoic acid (salicylic acid) as the main product.
(c)- Reimer-Tiemann reaction.
Treatment of phenol with chloroform in the presence of aqueous or potassium hydroxide at 340K followed by hydrolysis of the resulting product gives 2-hydroxybenzaldehyde (salicylaldehyde) as the major product.

(d)- Fittig reaction.
When only haloarenes are treated with sodium, diaryls are produced.

(e)- Carbylamine reaction.
Both aliphatic and aromatic primary amines when warmed with chloroform and an alcoholic solution of KOH produce isocyanides or carbylamines.

So, from the above observation, we can see that aniline is converted into phenyl isocyanide in the carbylamine reaction.
So, the correct answer is “Option E”.
Note: Don’t get confused between reamer-teimann and carbylamines reaction because both of them have chloroform as one of the reactants. In contrast, secondary and tertiary amines do not give the carbylamine reaction.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In order to find out the different types of gametes class 12 biology NEET_UG

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




