One mole of a perfect monoatomic gas is put through a cycle consisting of the following three reversible steps:
(CA): Isothermal compression from 2 atm and 10 litres to 20 atm and 1 litre.
(AB): Isobaric expansion to return the gas to the original volume of 10 litres with T going from ${T_1}$to ${T_2}$.
(BC): Cooling at constant volume to bring the gas to the original pressure and temperature.
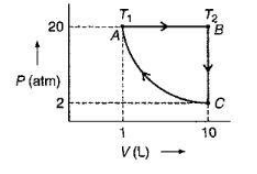
The steps are shown schematically in figure given above
(a) calculate ${T_1}$to ${T_2}$.
(b) Calculate $\Delta U,q$and$w$in calories, for each step and for the cycle.

Answer
602.4k+ views
Hint: For monoatomic gas, degrees of freedom is 3 $\therefore {C_v} = \dfrac{3}{2}R.$ We can use Ideal gas equation and the laws of thermodynamics to solve the given question.
Complete step by step answer:
Ideal gas equation, $PV = nRT$
First law of thermodynamics: $\Delta U = q + w$
We have $PV = nRT$
Where, P is pressure of ideal gas
V is volume of ideal gas
n is number of molecules
T is temperature
R is the ideal gas constant.
It is given that $n = 1$mole
In step CA:
Let us say, ${P_1}{V_1} = nR{T_1}$ . . . (1)
\[{P_1} = 2\] atm
${V_1} = 10$litres
$\therefore 2 \times 10 = 1 \times 0.821 \times {T_1}$
$\therefore {T_1} = 243.60K$
In step BC:
Volume is constant
$ \Rightarrow {P_2}{V_1} = 1R{T_2}$ …. (2)
Divide equation (1) by equation (2)
$\dfrac{{{P_1}{V_1}}}{{{P_2}{V_1}}} = \dfrac{{R{T_1}}}{{R{T_2}}}$
$ \Rightarrow \dfrac{{{P_1}}}{{{P_2}}} = \dfrac{{{T_1}}}{{{T_2}}}$
$\dfrac{2}{{20}} = \dfrac{{243.60}}{{{T_2}}}$
${T_2} = 10 \times 243.6$
$ = 2436K$
Now we have to find $\Delta U$i.e. change in energy
1) From A to B
${\left( {\Delta U} \right)_{AB}} = n{C_v}\Delta T$
$ = 1 \times \dfrac{3}{2}R({T_2} - {T_1})$
$ = 1 \times \dfrac{3}{2} \times 2 \times (246 - 243.6)$$\because R = 2$
$ = 6577.2cal$
2) From B to C
${\left( {\Delta U} \right)_{BC}} = n{C_v}R({T_1} - {T_2})$
$ = 1 \times \dfrac{3}{2} \times 2 \times (243.6 - 2436)$
$ = - 3577.2cal$
3) Process C to A is isothermal process
$\therefore {\left( {\Delta U} \right)_{CA}} = n{C_v}({T_1} - {T_1})$ (As there is no change in temperature.)
$ = 0$
Now the $\Delta U$of cycle is
\[\Delta U = {\left( {\Delta U} \right)_{AB}} + {(\Delta U)_{BC}} + {(\Delta U)_{CA}}\]
$ = 6577.2 - 6577.2 + 0$
$ = 0$
$\therefore $Net internal energy of cycle is zero
Now calculating the work done
1) A to B is isobaric process, so work done is
${\left( W \right)_{AB}} = - P\Delta V$
$ = - P({V_B} - {V_A})$
$ = - 20(10 - 1)$
$ = 180L$ atm
$ = - 180 \times \dfrac{2}{{0.0821}}cal$
$ = - 4384.9cal$
2) Volume is constant from B to C.
$\therefore \Delta V = 0$i.e. change in volume is zero.
$ \Rightarrow {W_{BC}} = 0$
Now, for finding $q$we use, the ${1^{st}}$law of thermodynamics
$\Delta U = q + w$
$q = \Delta U - w$
1) For A to B
${q_{AB}} = \Delta {U_{AB}} - {W_{AB}}$
$ = 6577.2 - ( - 4384.9)$
$ = 10962.1cal$
2) For B to C
${q_{BC}} = \Delta {U_{BC}} - {W_{BC}}$
$ = - 6577.2 - 0$
$ = - 6577.2cal$
3) For C to a
${q_{CA}} = \Delta {U_{CA}} - {W_{CA}}$
$ = 0 - 1122$
$ = - 1122cal$
$\therefore $For total cycle
${q_{cycle}} = {q_{AB}} + {q_{BC}} + {q_{CA}}$
$ = 10962.1 - 6577.2 - 1122$
$ = 3262.9cal$
Note: Positive sign indicates increase in energy and negative sign indicates the decrease in energy. To solve such types of questions, we need to have a clear idea about in which process temperature is constant and in which process volume is constant etc.
Complete step by step answer:
Ideal gas equation, $PV = nRT$
First law of thermodynamics: $\Delta U = q + w$
We have $PV = nRT$
Where, P is pressure of ideal gas
V is volume of ideal gas
n is number of molecules
T is temperature
R is the ideal gas constant.
It is given that $n = 1$mole
In step CA:
Let us say, ${P_1}{V_1} = nR{T_1}$ . . . (1)
\[{P_1} = 2\] atm
${V_1} = 10$litres
$\therefore 2 \times 10 = 1 \times 0.821 \times {T_1}$
$\therefore {T_1} = 243.60K$
In step BC:
Volume is constant
$ \Rightarrow {P_2}{V_1} = 1R{T_2}$ …. (2)
Divide equation (1) by equation (2)
$\dfrac{{{P_1}{V_1}}}{{{P_2}{V_1}}} = \dfrac{{R{T_1}}}{{R{T_2}}}$
$ \Rightarrow \dfrac{{{P_1}}}{{{P_2}}} = \dfrac{{{T_1}}}{{{T_2}}}$
$\dfrac{2}{{20}} = \dfrac{{243.60}}{{{T_2}}}$
${T_2} = 10 \times 243.6$
$ = 2436K$
Now we have to find $\Delta U$i.e. change in energy
1) From A to B
${\left( {\Delta U} \right)_{AB}} = n{C_v}\Delta T$
$ = 1 \times \dfrac{3}{2}R({T_2} - {T_1})$
$ = 1 \times \dfrac{3}{2} \times 2 \times (246 - 243.6)$$\because R = 2$
$ = 6577.2cal$
2) From B to C
${\left( {\Delta U} \right)_{BC}} = n{C_v}R({T_1} - {T_2})$
$ = 1 \times \dfrac{3}{2} \times 2 \times (243.6 - 2436)$
$ = - 3577.2cal$
3) Process C to A is isothermal process
$\therefore {\left( {\Delta U} \right)_{CA}} = n{C_v}({T_1} - {T_1})$ (As there is no change in temperature.)
$ = 0$
Now the $\Delta U$of cycle is
\[\Delta U = {\left( {\Delta U} \right)_{AB}} + {(\Delta U)_{BC}} + {(\Delta U)_{CA}}\]
$ = 6577.2 - 6577.2 + 0$
$ = 0$
$\therefore $Net internal energy of cycle is zero
Now calculating the work done
1) A to B is isobaric process, so work done is
${\left( W \right)_{AB}} = - P\Delta V$
$ = - P({V_B} - {V_A})$
$ = - 20(10 - 1)$
$ = 180L$ atm
$ = - 180 \times \dfrac{2}{{0.0821}}cal$
$ = - 4384.9cal$
2) Volume is constant from B to C.
$\therefore \Delta V = 0$i.e. change in volume is zero.
$ \Rightarrow {W_{BC}} = 0$
Now, for finding $q$we use, the ${1^{st}}$law of thermodynamics
$\Delta U = q + w$
$q = \Delta U - w$
1) For A to B
${q_{AB}} = \Delta {U_{AB}} - {W_{AB}}$
$ = 6577.2 - ( - 4384.9)$
$ = 10962.1cal$
2) For B to C
${q_{BC}} = \Delta {U_{BC}} - {W_{BC}}$
$ = - 6577.2 - 0$
$ = - 6577.2cal$
3) For C to a
${q_{CA}} = \Delta {U_{CA}} - {W_{CA}}$
$ = 0 - 1122$
$ = - 1122cal$
$\therefore $For total cycle
${q_{cycle}} = {q_{AB}} + {q_{BC}} + {q_{CA}}$
$ = 10962.1 - 6577.2 - 1122$
$ = 3262.9cal$
Note: Positive sign indicates increase in energy and negative sign indicates the decrease in energy. To solve such types of questions, we need to have a clear idea about in which process temperature is constant and in which process volume is constant etc.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




