Name the simplest hydrocarbon.
Answer
641.7k+ views
Hint: Simplest hydrocarbon belongs to the saturated class of hydrocarbons having a single bond in their structure.
Complete answer:
In organic chemistry a hydrocarbon is a chemical compound which consists of carbon atoms and hydrogen atoms. These hydrocarbons occur naturally and they are the basis of natural gas, coal, crude oil and other energy resources.
Types of hydrocarbons:
Hydrocarbons are classified as saturated hydrocarbons, unsaturated hydrocarbon and aromatic hydrocarbons.
Saturated hydrocarbons are the simplest form of hydrocarbons they consists of single bond and are saturated with hydrogens, the formula for acyclic hydrocarbon(open chain compound) i.e alkanes is \[{{\text{C}}_n}{{\text{H}}_{2n + 2}}\] where ${\text{n}}$ is the number of carbon atom.
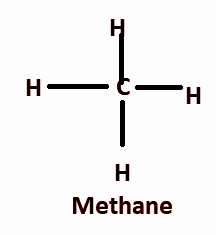
And methane is the simplest hydrocarbon which contains one carbon bonded to four hydrogen atoms.
Unsaturated hydrocarbons consists of one or more double bonds and triple bonds between the carbon atoms, those having double bonds they are called as alkenes and they have the formula \[{{\text{C}}_n}{{\text{H}}_{2n}}\] and those having triple bonds they are called as alkynes and they have the formula \[{{\text{C}}_n}{{\text{H}}_{2n - 2}}\].
Third one are aromatic hydrocarbons, they are also known as arenes(having a sigma bond and delocalized pi electrons between carbon atoms forming a circle) and they have at least one aromatic ring or we can say cyclic ring in their structure.
Note: Methane is the simplest alkane and alkanes have the formula \[{{\text{C}}_n}{{\text{H}}_{2n + 2}}\] where ${\text{n}}$ is the number of carbon atom, so methane contains one carbon atom and the number of hydrogen atoms are four i.e. \[{{\text{C}}_1}{{\text{H}}_{2 \times 1 + 2}} = {\text{C}}{{\text{H}}_4}\].
Complete answer:
In organic chemistry a hydrocarbon is a chemical compound which consists of carbon atoms and hydrogen atoms. These hydrocarbons occur naturally and they are the basis of natural gas, coal, crude oil and other energy resources.
Types of hydrocarbons:
Hydrocarbons are classified as saturated hydrocarbons, unsaturated hydrocarbon and aromatic hydrocarbons.
Saturated hydrocarbons are the simplest form of hydrocarbons they consists of single bond and are saturated with hydrogens, the formula for acyclic hydrocarbon(open chain compound) i.e alkanes is \[{{\text{C}}_n}{{\text{H}}_{2n + 2}}\] where ${\text{n}}$ is the number of carbon atom.
And methane is the simplest hydrocarbon which contains one carbon bonded to four hydrogen atoms.

Unsaturated hydrocarbons consists of one or more double bonds and triple bonds between the carbon atoms, those having double bonds they are called as alkenes and they have the formula \[{{\text{C}}_n}{{\text{H}}_{2n}}\] and those having triple bonds they are called as alkynes and they have the formula \[{{\text{C}}_n}{{\text{H}}_{2n - 2}}\].
Third one are aromatic hydrocarbons, they are also known as arenes(having a sigma bond and delocalized pi electrons between carbon atoms forming a circle) and they have at least one aromatic ring or we can say cyclic ring in their structure.
Note: Methane is the simplest alkane and alkanes have the formula \[{{\text{C}}_n}{{\text{H}}_{2n + 2}}\] where ${\text{n}}$ is the number of carbon atom, so methane contains one carbon atom and the number of hydrogen atoms are four i.e. \[{{\text{C}}_1}{{\text{H}}_{2 \times 1 + 2}} = {\text{C}}{{\text{H}}_4}\].
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




