Name the scientist who first formulated the atomic structure.
Answer
607.2k+ views
Hint: The atomic structure consists of Atomic particles. Atoms consist of three basic particles, protons, electrons and neutrons. The nucleus (center) of an atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and they contain the electrons.
Complete step by step answer:
-The atomic structure of an element refers to the constitution of its nucleus and the arrangement of the electrons around it. Primarily, the atomic structure of matter is made up of protons, electrons and neutrons.
-But, the atoms of different elements have different atomic structures because they contain different numbers of protons and electrons.
-The first scientific theory of atomic structure was proposed by John Dalton in1800s. The atomic structure is as shown-
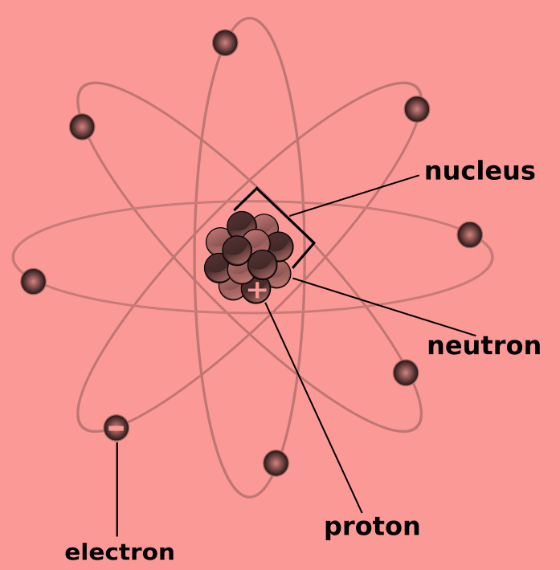
John Dalton suggested that all matter is made up of atoms, which were invisible and indestructible. He also stated that all the atoms of an element were exactly the same, but the atoms of different elements differ in size and mass.
Note:
There were few postulates of this theory:
-Every matter is made up of atoms.
-Atoms are invisible.
-Specific elements have only one type of atom in them.
-Each atom has its own constant mass that varies from element to element.
-Atoms undergo rearrangement during a chemical reaction.
Complete step by step answer:
-The atomic structure of an element refers to the constitution of its nucleus and the arrangement of the electrons around it. Primarily, the atomic structure of matter is made up of protons, electrons and neutrons.
-But, the atoms of different elements have different atomic structures because they contain different numbers of protons and electrons.
-The first scientific theory of atomic structure was proposed by John Dalton in1800s. The atomic structure is as shown-

John Dalton suggested that all matter is made up of atoms, which were invisible and indestructible. He also stated that all the atoms of an element were exactly the same, but the atoms of different elements differ in size and mass.
Note:
There were few postulates of this theory:
-Every matter is made up of atoms.
-Atoms are invisible.
-Specific elements have only one type of atom in them.
-Each atom has its own constant mass that varies from element to element.
-Atoms undergo rearrangement during a chemical reaction.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




