What is the molecular shape of hydrogen sulphide molecule ${H_2}S$?
Answer
544.5k+ views
Hint: One of the most important theory which is used to predict the molecular shape and geometry of a molecule is valence shell electron pair repulsion theory i.e., VSEPR theory which is based on a concept that the lone pairs and bond pairs in a molecule will be arranged in such a manner that the repulsive forces are minimum.
Complete answer:
According to VSEPR theory,
1.In a polyatomic molecule i.e., a molecule which consists of three or more atoms, must have a central atom and all other atoms present in the molecule will form a bond with that central atom.
2.The total number of valence electrons plays a major role in deciding the shape of the molecule.
3.The lone pair of electrons on an atom has the tendency to arrange itself in such a way, that the repulsion is minimum.
4.If the central atom consists of both lone pairs as well as bond pairs, then the resulting shape of the molecule will be distorted.
5.The geometry of the molecule is decided by its steric number.
Therefore, for hydrogen sulphide molecule i.e., ${H_2}S$
Steric number $ = 4$
Corresponding hybridized state $ = s{p^3}$
Expected geometry is tetrahedral for the given case, but due to presence of two lone pair of electrons the geometry is distorted and the molecule forms a bent shape structure as follows:
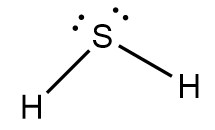
Hence, the molecular shape of hydrogen sulphide is bent.
Note:
It is important to note that the number of atoms, groups and lone pairs connected to the central atom together make up the steric number of the molecule. In simple words, steric number is the summation of the number of lone pairs on a central atom and number of atoms connected to it via a single bond. For example, in ${H_2}S$, there are two bond pairs and two lone pairs. Therefore, the steric number of ${H_2}S$ is four.
Complete answer:
According to VSEPR theory,
1.In a polyatomic molecule i.e., a molecule which consists of three or more atoms, must have a central atom and all other atoms present in the molecule will form a bond with that central atom.
2.The total number of valence electrons plays a major role in deciding the shape of the molecule.
3.The lone pair of electrons on an atom has the tendency to arrange itself in such a way, that the repulsion is minimum.
4.If the central atom consists of both lone pairs as well as bond pairs, then the resulting shape of the molecule will be distorted.
5.The geometry of the molecule is decided by its steric number.
Therefore, for hydrogen sulphide molecule i.e., ${H_2}S$
Steric number $ = 4$
Corresponding hybridized state $ = s{p^3}$
Expected geometry is tetrahedral for the given case, but due to presence of two lone pair of electrons the geometry is distorted and the molecule forms a bent shape structure as follows:

Hence, the molecular shape of hydrogen sulphide is bent.
Note:
It is important to note that the number of atoms, groups and lone pairs connected to the central atom together make up the steric number of the molecule. In simple words, steric number is the summation of the number of lone pairs on a central atom and number of atoms connected to it via a single bond. For example, in ${H_2}S$, there are two bond pairs and two lone pairs. Therefore, the steric number of ${H_2}S$ is four.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




