What is meant by
i.) peptide linkage
ii.) biocatalysts
Answer
623.7k+ views
Hint: Peptide linkage is a bond formed between two amino acids during protein formation.
Biocatalysts are the natural molecules which help in increase in rate of reaction.
Complete step by step answer:
Peptide linkage :- It is the name given to a chemical bond formed between C-N when proteins are formed from amino acids. When two molecules combine; the carboxyl group of one molecule reacts with the amino group of the other molecule resulting in formation of a molecule of water. It is a dehydration reaction. The reaction generally occurs between two amino acids.
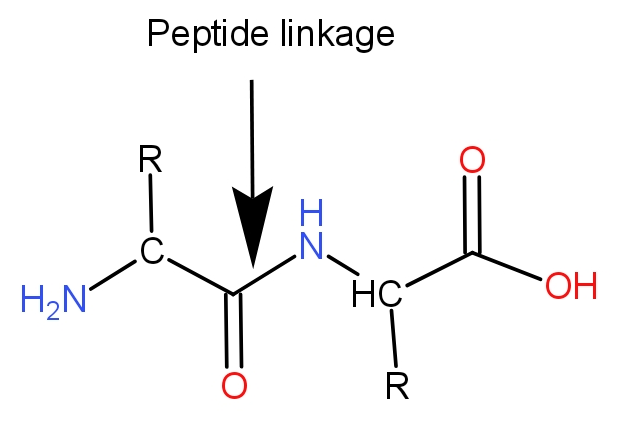
This bond is covalent in nature. The monomers i.e. amino acids are joined forming a polymer which is protein.
Biocatalysts :- These are defined as the natural molecules that increase the rate of chemical reactions in the cells of living beings. These natural molecules can be enzymes or any other living small micro-organism or its part which performs chemical transformations on organic compounds in the organism. The enzymes are known as protein catalysts because they are majorly made up of proteins.
Biocatalysts provide several advantages over inorganic catalysts. The main one could be that these work in mild conditions. Thus, there are no chances of side reactions and are thus less harmful.
Note:
Peptide linkage is also a type of condensation reaction.
The formation of the peptide bond requires energy which is derived from ATP in living beings. The process is normally catalysed by enzymes which are known as peptidases or proteases. These enzymes are called biocatalysts.
The peptide bond is not so reactive because of resonance stabilization.
Biocatalysts are the natural molecules which help in increase in rate of reaction.
Complete step by step answer:
Peptide linkage :- It is the name given to a chemical bond formed between C-N when proteins are formed from amino acids. When two molecules combine; the carboxyl group of one molecule reacts with the amino group of the other molecule resulting in formation of a molecule of water. It is a dehydration reaction. The reaction generally occurs between two amino acids.

This bond is covalent in nature. The monomers i.e. amino acids are joined forming a polymer which is protein.
Biocatalysts :- These are defined as the natural molecules that increase the rate of chemical reactions in the cells of living beings. These natural molecules can be enzymes or any other living small micro-organism or its part which performs chemical transformations on organic compounds in the organism. The enzymes are known as protein catalysts because they are majorly made up of proteins.
Biocatalysts provide several advantages over inorganic catalysts. The main one could be that these work in mild conditions. Thus, there are no chances of side reactions and are thus less harmful.
Note:
Peptide linkage is also a type of condensation reaction.
The formation of the peptide bond requires energy which is derived from ATP in living beings. The process is normally catalysed by enzymes which are known as peptidases or proteases. These enzymes are called biocatalysts.
The peptide bond is not so reactive because of resonance stabilization.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




