LPG used as domestic fuel is mixed with a strong-smelling liquid for---------.
(A) Better combustion
(B) Good small/fragrance
(C) Detection of gas leakage
(D) Higher calorific value
Answer
623.4k+ views
Hint: Generally a fuel gas does not have any odour. LPG is a mixture of propane and butane and it does not have any odour. By adding a strong-smelling liquid called Ethyl Mercaptan LPG gives a strong smell of rotten cabbages.
Complete step by step solution:
-Generally fuel gases like LPG supplied for domestic use do not have an odour.
-If there is any leak of LPG we cannot find it because we cannot see the gases and there is no odour to LPG.
-So, to detect the leakage of LPG gas a strong-smelling chemical called Ethyl Mercaptan is added.
-After adding Ethyl Mercaptan to LPG we can detect the leakage very easily.
-So, the correct option is C, detection of gas leakage.
Additional information:
-Ethyl Mercaptan is a colourless gas and has a low boiling point.
-The molecular formula of Ethyl Mercaptan is\[{{C}_{2}}{{H}_{6}}S\].
-Ethyl Mercaptan is also called Ethanethiol.
-The structure of Ethyl Mercaptan is as follows.
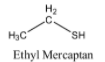
-Due to the presence of sulphur Ethyl Mercaptan is too smelly.
-Generally Sulphur compounds are smelly in nature.
Note:-Ethyl Mercaptan is only added to LPG gas (used for the domestic purpose), not to CNG, which is used to run vehicles.
LPG – Liquefied Petroleum Gas
CNG – Compressed Natural Gas (composed of methane gas)
-By adding Ethyl Mercaptan to LPG the calorific value of LPG is not going to affect.
Complete step by step solution:
-Generally fuel gases like LPG supplied for domestic use do not have an odour.
-If there is any leak of LPG we cannot find it because we cannot see the gases and there is no odour to LPG.
-So, to detect the leakage of LPG gas a strong-smelling chemical called Ethyl Mercaptan is added.
-After adding Ethyl Mercaptan to LPG we can detect the leakage very easily.
-So, the correct option is C, detection of gas leakage.
Additional information:
-Ethyl Mercaptan is a colourless gas and has a low boiling point.
-The molecular formula of Ethyl Mercaptan is\[{{C}_{2}}{{H}_{6}}S\].
-Ethyl Mercaptan is also called Ethanethiol.
-The structure of Ethyl Mercaptan is as follows.

-Due to the presence of sulphur Ethyl Mercaptan is too smelly.
-Generally Sulphur compounds are smelly in nature.
Note:-Ethyl Mercaptan is only added to LPG gas (used for the domestic purpose), not to CNG, which is used to run vehicles.
LPG – Liquefied Petroleum Gas
CNG – Compressed Natural Gas (composed of methane gas)
-By adding Ethyl Mercaptan to LPG the calorific value of LPG is not going to affect.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources

Which Country Has the Largest Border with India?

Choose the feminine form of the given noun Fox AFoxess class 10 english CBSE




