Inorganic graphite is:
(A). \[{\left( {BN} \right)_n}\]
(B) $B{F_4}$
(C) ${B_2}{H_6}$
(D) ${B_2}{N_2}{H_6}$
Answer
612.3k+ views
Hint: Inorganic graphite is an inorganic compound whose structure is like graphite with alternating atoms replacing the carbon atoms in hexagonal structure.
Complete step by step answer:
Boron nitride is sometimes referred to as ‘inorganic graphite’ because its structure is like graphite with alternating boron and nitrogen atoms replacing the carbon atoms in the hexagonal structure.
$BN$ is isoelectronic to a similarly structured carbon lattice and thus exists in various crystalline forms.
The structure of $BN$ is
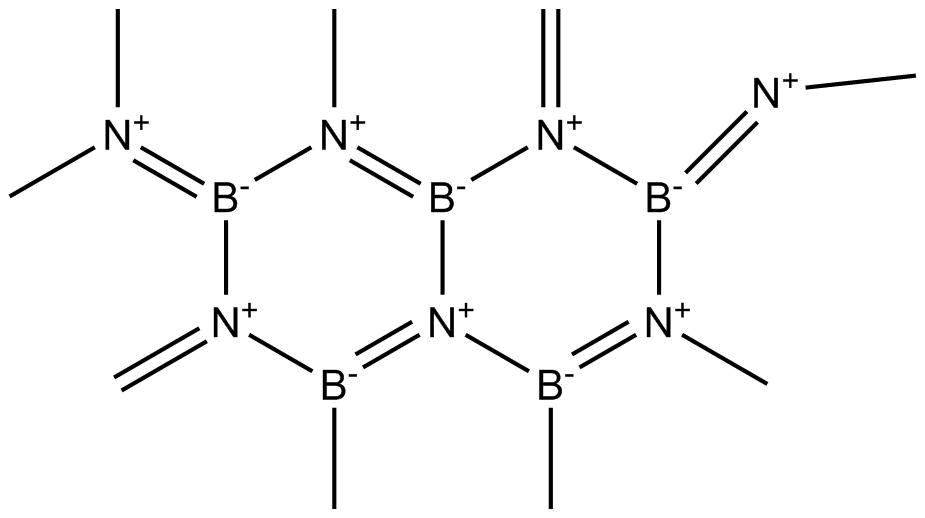
Figure-The structure of Boron Nitride
Additional information:
Boron nitride is produced synthetically. Hexagonal boron nitride is obtained by the reacting trioxide $\left( {{B_2}{O_3}} \right)$ or boric acid $\left( {{H_3}B{O_3}} \right)$ with ammonia ${\left( {NH} \right)_3}$ or $\left( {N{H_2}CON{H_2}} \right)$ in a nitrogen atmosphere.
${B_2}{O_3} + 2N{H_3} \to 2BN + 3{H_2}O$ $\left( {T = 900^\circ c} \right)$
$B{\left( {OH} \right)_3} + N{H_3} \to BN + 3{H_2}O$ $\left( {T = 900^\circ c} \right)$
${B_2}{O_3} + CO{\left( {N{H_2}} \right)_2} \to 2BN + C{O_2} + 2{H_2}O$ $\left( {T > 1000^\circ c} \right)$
${B_2}{O_3} + 3Ca{B_6} + IO{N_2} \to 2OBN + 3CaO$ $\left( {T > 1500^\circ c} \right)$
Similar to graphite, various molecules such as $N{H_3}$ or alkali metals can be intercalated into hexagonal boron nitride, that is inserted between its layers. Both experiment and theory suggest the intercalation is much more difficult for $BN$ than for graphite.
Note:
$BN$ is the most widely used polymorph. It is a good lubricant at both low and high temperature. $BN$ is used as an lubricant when electrical conductivity or chemical reactivity of graphite would be problematic.
Complete step by step answer:
Boron nitride is sometimes referred to as ‘inorganic graphite’ because its structure is like graphite with alternating boron and nitrogen atoms replacing the carbon atoms in the hexagonal structure.
$BN$ is isoelectronic to a similarly structured carbon lattice and thus exists in various crystalline forms.
The structure of $BN$ is

Figure-The structure of Boron Nitride
Additional information:
Boron nitride is produced synthetically. Hexagonal boron nitride is obtained by the reacting trioxide $\left( {{B_2}{O_3}} \right)$ or boric acid $\left( {{H_3}B{O_3}} \right)$ with ammonia ${\left( {NH} \right)_3}$ or $\left( {N{H_2}CON{H_2}} \right)$ in a nitrogen atmosphere.
${B_2}{O_3} + 2N{H_3} \to 2BN + 3{H_2}O$ $\left( {T = 900^\circ c} \right)$
$B{\left( {OH} \right)_3} + N{H_3} \to BN + 3{H_2}O$ $\left( {T = 900^\circ c} \right)$
${B_2}{O_3} + CO{\left( {N{H_2}} \right)_2} \to 2BN + C{O_2} + 2{H_2}O$ $\left( {T > 1000^\circ c} \right)$
${B_2}{O_3} + 3Ca{B_6} + IO{N_2} \to 2OBN + 3CaO$ $\left( {T > 1500^\circ c} \right)$
Similar to graphite, various molecules such as $N{H_3}$ or alkali metals can be intercalated into hexagonal boron nitride, that is inserted between its layers. Both experiment and theory suggest the intercalation is much more difficult for $BN$ than for graphite.
Note:
$BN$ is the most widely used polymorph. It is a good lubricant at both low and high temperature. $BN$ is used as an lubricant when electrical conductivity or chemical reactivity of graphite would be problematic.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

10 examples of friction in our daily life




