In the following reaction which of the following products can not form?
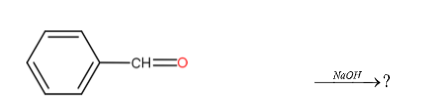
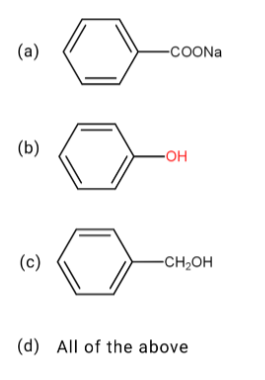
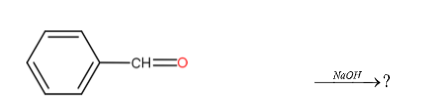

Answer
588.3k+ views
Hint: When we use condensed aldehydes or ketones in presence of sodium hydroxide there can only be two reactions that take place: aldol condensation or Cannizzaro reaction, depending upon the factors we should choose one.
Complete Step by step answer: So first of all we should identify which condensation reaction takes place here. Let us look into its $\alpha $-hydrogen of benzaldehyde, we can see the $\alpha $-carbon atom is attached to its neighbouring benzene carbons hence there is no space to accommodate the $\alpha $-hydrogen atom. So we can say that there is no $\alpha $-hydrogen atom in benzaldehyde. Therefore, the reaction will be Cannizzaro reaction.
It is a self oxidation-reduction reaction in which aldehydes that do not have any $\alpha $-hydrogen atom undergo disproportionation reaction (i.e., self-redox reaction) in the presence of 50% aqueous or ethanolic solution of alkali in which one of the molecules being reduced to alcohol and other being oxidised to the salt of the corresponding acid. So when benzaldehyde is reacted with NaOH, we get two substance one of them will its alcohol compound and the other one is its salt of corresponding acid.
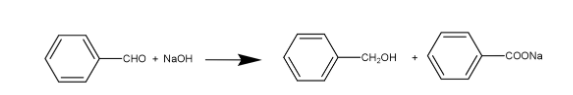
We can see that when benzaldehyde is treated with NaOH we get the two compounds. One is the reduced one to its corresponding alcohol i.e., benzyl alcohol and the other one is its oxidised form i.e., sodium benzoate.
It is clearly evident that phenol is not obtained from the above reaction.
Therefore, option (b) is correct.
Note: We cannot produce phenol from Cannizzaro reaction since the reaction happens at the functional group it cannot attack the benzene ring, that is why the phenol is not obtained from this reaction. It may come to our mind that the hydroxyl group may attach to the benzene ring, but it does not happen here.
Complete Step by step answer: So first of all we should identify which condensation reaction takes place here. Let us look into its $\alpha $-hydrogen of benzaldehyde, we can see the $\alpha $-carbon atom is attached to its neighbouring benzene carbons hence there is no space to accommodate the $\alpha $-hydrogen atom. So we can say that there is no $\alpha $-hydrogen atom in benzaldehyde. Therefore, the reaction will be Cannizzaro reaction.
It is a self oxidation-reduction reaction in which aldehydes that do not have any $\alpha $-hydrogen atom undergo disproportionation reaction (i.e., self-redox reaction) in the presence of 50% aqueous or ethanolic solution of alkali in which one of the molecules being reduced to alcohol and other being oxidised to the salt of the corresponding acid. So when benzaldehyde is reacted with NaOH, we get two substance one of them will its alcohol compound and the other one is its salt of corresponding acid.

We can see that when benzaldehyde is treated with NaOH we get the two compounds. One is the reduced one to its corresponding alcohol i.e., benzyl alcohol and the other one is its oxidised form i.e., sodium benzoate.
It is clearly evident that phenol is not obtained from the above reaction.
Therefore, option (b) is correct.
Note: We cannot produce phenol from Cannizzaro reaction since the reaction happens at the functional group it cannot attack the benzene ring, that is why the phenol is not obtained from this reaction. It may come to our mind that the hydroxyl group may attach to the benzene ring, but it does not happen here.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE




