In the following reaction, ${{\text{C}}_{6}}{{\text{H}}_{5}}\text{CHO + C}{{\text{H}}_{3}}\text{COC}{{\text{H}}_{3}}\,\xrightarrow[\Delta ]{\text{O}{{\text{H}}^{-}}}\text{ P}$ which product is formed?
A. ${{\text{C}}_{6}}{{\text{H}}_{5}}\text{COH}$
B. ${{\text{C}}_{6}}{{\text{H}}_{5}}\text{-CO-}{{\text{C}}_{6}}{{\text{H}}_{5}}$
C. ${{\text{C}}_{6}}{{\text{H}}_{5}}\text{CH=CHCOC}{{\text{H}}_{3}}$
D. ${{\text{C}}_{6}}{{\text{H}}_{5}}-\text{CO - CO - }{{\text{C}}_{6}}{{\text{H}}_{5}}$
Answer
612k+ views
Hint: The reaction is an example of an aldol condensation. Aldol condensation is a reaction in which the carbonyl group reacts with the enol group yielding beta-hydroxy aldehyde/ ketone. This is a type of condensation reaction.
Complete answer:
-In the given reaction, the reactants are aldehyde and ketone and we have to find the product P formed when they both will react.
-They both will undergo aldol condensation reaction in which the following steps are followed:
Step I: firstly, the hydroxyl ion will deprotonate the aldehyde group by breaking the bond between carbon and hydrogen and releases enol ion and water i.e.
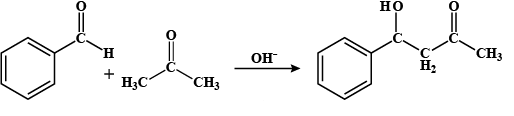
Benzaldehyde Acetone 4 - hydroxy -4-phenyl but-2-one
Step II: Now, after deprotonation, the ketone attaches to the aldehyde group and forms 4 - hydroxy -4-phenyl but-2-one
Step III: Now, it will be heated and undergoes a dehydration process in which the water is released, by removal of a hydroxyl group from benzaldehyde and hydrogen from ketone that is attached to the benzaldehyde.

4 - hydroxy -4-phenyl but-2-one 4-phenyl but-3-ene-2-one
-So, the product P formed is ${{\text{C}}_{6}}{{\text{H}}_{5}}\text{CH=CHCOC}{{\text{H}}_{3}}$.
So, the correct answer is “Option C”.
Note: For an aldol condensation to take place the compound should have an alpha-hydrogen i.e. hydrogen which is attached directly to the functional group. Crossed aldol condensation is the reaction in which the two different aldehyde or ketone compounds react with each other.
.
Complete answer:
-In the given reaction, the reactants are aldehyde and ketone and we have to find the product P formed when they both will react.
-They both will undergo aldol condensation reaction in which the following steps are followed:
Step I: firstly, the hydroxyl ion will deprotonate the aldehyde group by breaking the bond between carbon and hydrogen and releases enol ion and water i.e.

Benzaldehyde Acetone 4 - hydroxy -4-phenyl but-2-one
Step II: Now, after deprotonation, the ketone attaches to the aldehyde group and forms 4 - hydroxy -4-phenyl but-2-one
Step III: Now, it will be heated and undergoes a dehydration process in which the water is released, by removal of a hydroxyl group from benzaldehyde and hydrogen from ketone that is attached to the benzaldehyde.

4 - hydroxy -4-phenyl but-2-one 4-phenyl but-3-ene-2-one
-So, the product P formed is ${{\text{C}}_{6}}{{\text{H}}_{5}}\text{CH=CHCOC}{{\text{H}}_{3}}$.
So, the correct answer is “Option C”.
Note: For an aldol condensation to take place the compound should have an alpha-hydrogen i.e. hydrogen which is attached directly to the functional group. Crossed aldol condensation is the reaction in which the two different aldehyde or ketone compounds react with each other.
.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




