In $ICl_4^ \ominus $ the shape is square planar. The number of bond pair-lone repulsion at $90^\circ $ are:
(A). $6$
(B). $8$
(C). $12$
(D). $4$
Answer
593.4k+ views
Hint:
We know that $ICl_4^ \ominus $ is $A{B_4}{E_2}$ type molecule and it shows:
Electronic Structure: $A{B_4}{E_2}$
Electronic Geometry: Octahedral
Hybridization of central atom is $s{p^3}{d^2}$
Complete step by step answer:
There are two lone pairs of electrons which are perpendicular to the square plane. In the square plane there are $8$ lone pair electrons.
Thus, repulsion at $90^\circ = 8$

Therefore, the correct option is (B) $8$.
Additional Information:
In the lewis structure of $ICl_4^ \ominus $ there are total $36$ valence electrons.
Since Iodine $\left( I \right)$ is below period $3$ on the periodic table, it can have more than $8$ electrons. In the lewis structure of $ICl_4^ \ominus $ the iodine atom has $12$ valence electrons.
$3 - D$ structure.
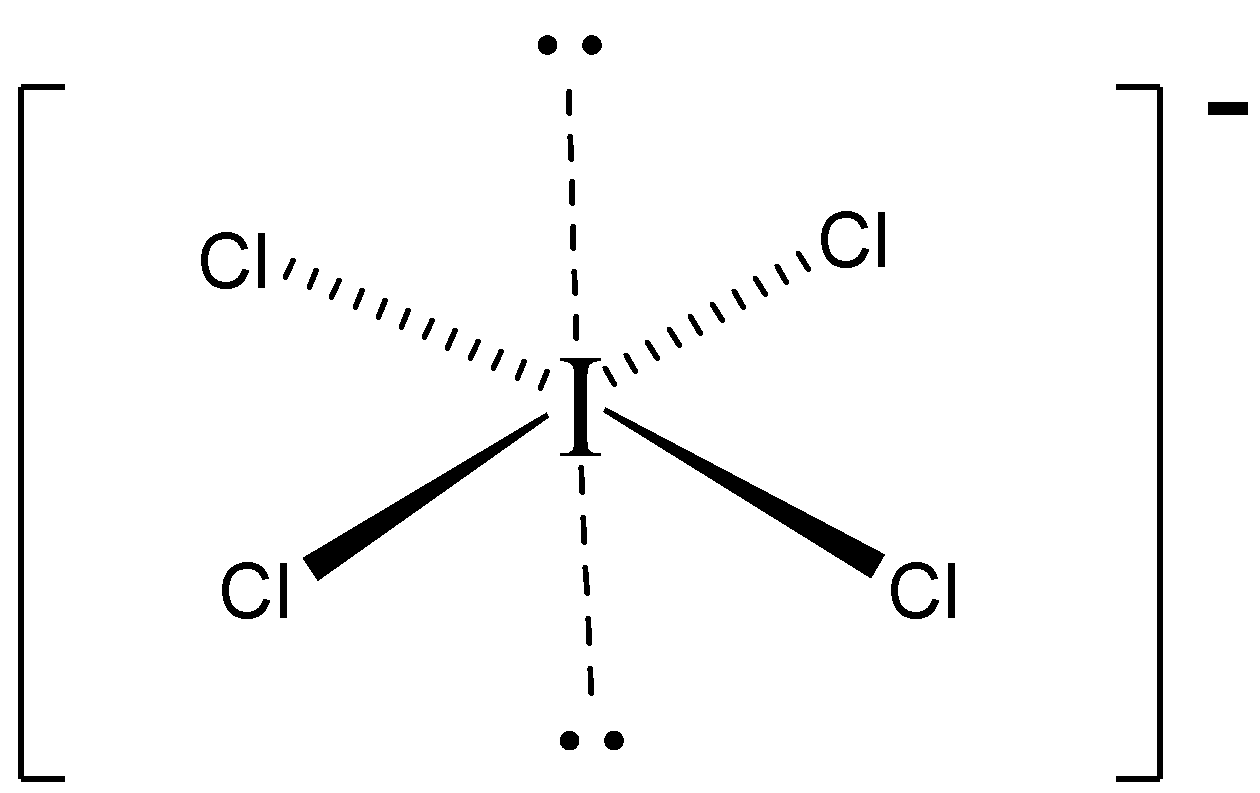
Important Points:
In $ICl_4^ \ominus $ lewis structure, Iodine $\left( I \right)$ has the least electronegativity and goes in the center of lewis structure.
The $ICl_4^ \ominus $ lewis structure you’ll need to put more than eight electrons on the iodine atom.
In the lewis structure for $ICl_4^ \ominus $, there are a total of $36$ electrons.
Note: Note that you should put $ICl_4^ \ominus $ lewis structure in brackets with $ - 1$ charge outside to show that it is an ion with negative one charge.
We know that $ICl_4^ \ominus $ is $A{B_4}{E_2}$ type molecule and it shows:
Electronic Structure: $A{B_4}{E_2}$
Electronic Geometry: Octahedral
Hybridization of central atom is $s{p^3}{d^2}$
Complete step by step answer:
There are two lone pairs of electrons which are perpendicular to the square plane. In the square plane there are $8$ lone pair electrons.
Thus, repulsion at $90^\circ = 8$

Therefore, the correct option is (B) $8$.
Additional Information:
In the lewis structure of $ICl_4^ \ominus $ there are total $36$ valence electrons.
Since Iodine $\left( I \right)$ is below period $3$ on the periodic table, it can have more than $8$ electrons. In the lewis structure of $ICl_4^ \ominus $ the iodine atom has $12$ valence electrons.
$3 - D$ structure.

Important Points:
In $ICl_4^ \ominus $ lewis structure, Iodine $\left( I \right)$ has the least electronegativity and goes in the center of lewis structure.
The $ICl_4^ \ominus $ lewis structure you’ll need to put more than eight electrons on the iodine atom.
In the lewis structure for $ICl_4^ \ominus $, there are a total of $36$ electrons.
Note: Note that you should put $ICl_4^ \ominus $ lewis structure in brackets with $ - 1$ charge outside to show that it is an ion with negative one charge.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




