In $\text{Fe}{{\left( \text{CO} \right)}_{5}}$, the $\text{Fe}-\text{C}$ bond possess:
A. $\pi $ character only
B. both $\pi $ and $\sigma $ characters
C. ionic character
D. $\sigma $ character only
Answer
614.7k+ views
Hint: The $\text{Fe}{{\left( \text{CO} \right)}_{5}}$ compound forms metal-carbonyl bonds with each other. The structure of this compound is the same as others but the bonding is different. This bonding involves the d-orbitals and antibonding orbitals of the metal atom.
Complete step by step answer:
- In $\text{Fe}{{\left( \text{CO} \right)}_{5}}$, there are same ligands which is $\text{CO}$ so, $\text{Fe}{{\left( \text{CO} \right)}_{5}}$ is a homoleptic carbonyl compound. Such compounds are formed by most of the transition metals or d-block elements and they have simple and well defined structures. The shape of the compound is trigonal bi-pyramidal. The IUPAC name of the compound is Pentacarbonyliron $\left( 0 \right)$. The structure is
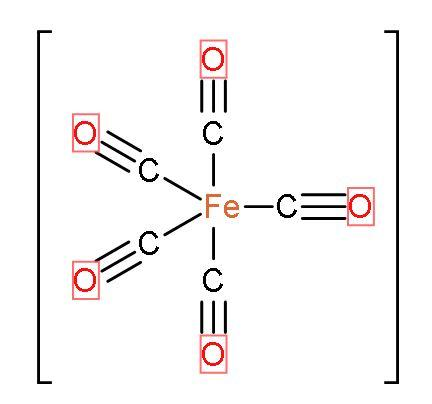
Let us discuss the special type of bonding in $\text{Fe}{{\left( \text{CO} \right)}_{5}}$:
The special type of bonding is named as ‘synergic bonding’. The ligand $\left( \text{CO} \right)$ donates its lone pair of electrons to the vacant orbitals of the central metal atom which is an iron atom and forms the sigma-bond with it. As the iron atom also possesses some electrons in its d-orbitals. The configuration of iron is $1{{\text{s}}^{2}}\text{2}{{\text{s}}^{2}}\text{2}{{\text{p}}^{6}}\text{3}{{\text{s}}^{2}}\text{3}{{\text{p}}^{6}}\text{4}{{\text{s}}^{2}}\text{3}{{\text{d}}^{6}}$ or $\begin{matrix}
\uparrow \downarrow & \uparrow & \uparrow & \uparrow & \uparrow \\
\end{matrix}$.
It back donates these electrons to the molecular orbitals of the ligand forming a $\pi $-bond. The $\text{M}-\text{C}$ $\pi $ bond is formed by the donation of electrons from a filled d-orbital of metal into the vacant antibonding ${{\pi }^{*}}$ orbital of carbon monoxide. The bonding looks like
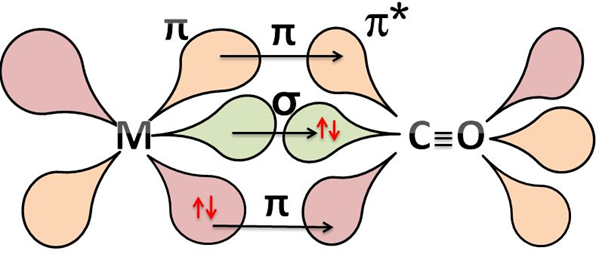
This creates a synergic effect between the metal to ligand which strengthens the bond between $\text{CO}$ and the metal. The metal-carbon bond in metal carbonyls possesses both $\sigma $ and $\pi $ character.
So, the correct answer is “Option B”.
Note: One important point to keep in mind is that the metal atom donates its electron pairs to the antibonding MO of $\text{CO}$, so the $\text{CO}$ bond is weakened by this synergic bonding which leads to a larger $\text{CO}$ bond length in the complex.
Complete step by step answer:
- In $\text{Fe}{{\left( \text{CO} \right)}_{5}}$, there are same ligands which is $\text{CO}$ so, $\text{Fe}{{\left( \text{CO} \right)}_{5}}$ is a homoleptic carbonyl compound. Such compounds are formed by most of the transition metals or d-block elements and they have simple and well defined structures. The shape of the compound is trigonal bi-pyramidal. The IUPAC name of the compound is Pentacarbonyliron $\left( 0 \right)$. The structure is

Let us discuss the special type of bonding in $\text{Fe}{{\left( \text{CO} \right)}_{5}}$:
The special type of bonding is named as ‘synergic bonding’. The ligand $\left( \text{CO} \right)$ donates its lone pair of electrons to the vacant orbitals of the central metal atom which is an iron atom and forms the sigma-bond with it. As the iron atom also possesses some electrons in its d-orbitals. The configuration of iron is $1{{\text{s}}^{2}}\text{2}{{\text{s}}^{2}}\text{2}{{\text{p}}^{6}}\text{3}{{\text{s}}^{2}}\text{3}{{\text{p}}^{6}}\text{4}{{\text{s}}^{2}}\text{3}{{\text{d}}^{6}}$ or $\begin{matrix}
\uparrow \downarrow & \uparrow & \uparrow & \uparrow & \uparrow \\
\end{matrix}$.
It back donates these electrons to the molecular orbitals of the ligand forming a $\pi $-bond. The $\text{M}-\text{C}$ $\pi $ bond is formed by the donation of electrons from a filled d-orbital of metal into the vacant antibonding ${{\pi }^{*}}$ orbital of carbon monoxide. The bonding looks like

This creates a synergic effect between the metal to ligand which strengthens the bond between $\text{CO}$ and the metal. The metal-carbon bond in metal carbonyls possesses both $\sigma $ and $\pi $ character.
So, the correct answer is “Option B”.
Note: One important point to keep in mind is that the metal atom donates its electron pairs to the antibonding MO of $\text{CO}$, so the $\text{CO}$ bond is weakened by this synergic bonding which leads to a larger $\text{CO}$ bond length in the complex.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE




