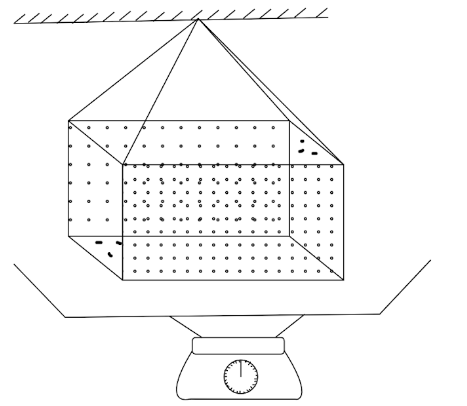
In ancient times, a weight thermometer was used to measure the temperature. The apparatus is shown in figure. The container's volume at absolute temperature is ${V_0}$. ${\gamma _L}$ is the coefficient of volume expansion of the liquid filled. ${\gamma _C}$ is the coefficient of volume expansion of the metal of the container. Density of liquid at absolute temperature is ${\rho _{{L_0}}}$. If $\vartriangle m$ is the mass of liquid overflowing, then $\vartriangle T$ is given by
A. $\vartriangle T = \dfrac{{\vartriangle m}}{{{\rho _{{L_0}}}{V_0}{\gamma _L} - \vartriangle m{\gamma _L}}}$
B. $\vartriangle T = \dfrac{{\vartriangle m}}{{{\rho _{{L_0}}}{V_0}{\gamma _L} + \vartriangle m{\gamma _L}}}$
C. $\vartriangle T = \dfrac{{\vartriangle m}}{{{\rho _{{L_0}}}{V_0}({\gamma _L} - {\gamma _C}) - \vartriangle m{\gamma _L}}}$
D. none of these

Answer
528k+ views
Hint: We will find out the volume of the liquid that will overflow when there is any temperature change. After that we simply know that change in mass is equal to the change in velocity of the substance to its density. Then, by ordinary calculation we will be able to find a change in temperature.
Formula used:
${\rho _f}\left( {1 + \gamma \vartriangle T} \right) = {\rho _i}$
where $f$ implies final ang $i$ implies initial.
Also the formula for volume expansion is given as,
${V_f} = {V_i}\left( {1 + \gamma \vartriangle T} \right)$
where, ${V_i}$ is the final volume, ${V_i}$ is the initial volume and $\gamma $ is the coefficient of volume expansion.
Complete step by step answer:
In the above question it is mentioned that ${\gamma _L}$ is the coefficient of volume expansion of the liquid filled. ${\gamma _C}$ is the coefficient of volume expansion of the metal of the container. ${\rho _{{L_0}}}$ is the density of the liquid at absolute temperature.
${V_0}$ is the volume of the container. We know that,
${V_f} = {V_i}\left( {1 + \gamma \vartriangle T} \right)$ where, ${V_i}$ is the final volume, ${V_i}$ is the initial volume and $\gamma $ is the coefficient of volume expansion.
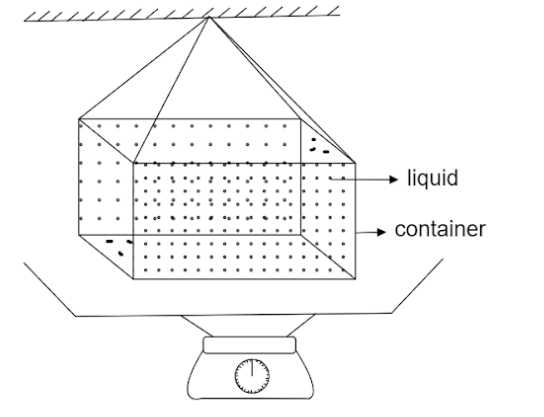
Thus from the above question we get,
${V_f} = {V_0}\left\{ {1 + \left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T} \right\}$
$\Rightarrow {V_f} - {V_0} = {V_0}\left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T$
Now, the change in volume is given as,
$\vartriangle V = {V_0}\left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T - - - - - \left( 1 \right)$
Again we should find the change in density,
Let ${\rho _L}$ be the final density after change of temperature $\vartriangle T$,
${\rho _L} = \dfrac{{{\rho _{{L_0}}}}}{{\left( {1 + {\gamma _L}\vartriangle T} \right)}} - - - - - \left( 2 \right)$
Now, the mass collected can be represented as,
$\vartriangle m = \vartriangle V \times {\rho _{{L_{}}}}$
Substituting the value of $\vartriangle V$ and ${\rho _L}$ from equation $\left( 1 \right)$ and $\left( 2 \right)$ we get,
$\vartriangle m = {V_0}\left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T \times \dfrac{{{\rho _{{L_0}}}}}{{\left( {1 + {\gamma _L}\vartriangle T} \right)}}$
Interchanging the variables we get,
$\therefore \vartriangle T = \dfrac{{\vartriangle m}}{{{\rho _{{L_0}}}{V_0}({\gamma _L} - {\gamma _C}) - \vartriangle m{\gamma _L}}}$
So, the correct option is C.
Note: There is a possibility to overlook the volumetric expansion of the liquid inside the container. It is most important to recall that the volumetric expansion of solids helps to expand the metal surface which in turn allows less liquid to overflow. It must be subtracted while finding the mass overflow as done in the solution.
Formula used:
${\rho _f}\left( {1 + \gamma \vartriangle T} \right) = {\rho _i}$
where $f$ implies final ang $i$ implies initial.
Also the formula for volume expansion is given as,
${V_f} = {V_i}\left( {1 + \gamma \vartriangle T} \right)$
where, ${V_i}$ is the final volume, ${V_i}$ is the initial volume and $\gamma $ is the coefficient of volume expansion.
Complete step by step answer:
In the above question it is mentioned that ${\gamma _L}$ is the coefficient of volume expansion of the liquid filled. ${\gamma _C}$ is the coefficient of volume expansion of the metal of the container. ${\rho _{{L_0}}}$ is the density of the liquid at absolute temperature.
${V_0}$ is the volume of the container. We know that,
${V_f} = {V_i}\left( {1 + \gamma \vartriangle T} \right)$ where, ${V_i}$ is the final volume, ${V_i}$ is the initial volume and $\gamma $ is the coefficient of volume expansion.

Thus from the above question we get,
${V_f} = {V_0}\left\{ {1 + \left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T} \right\}$
$\Rightarrow {V_f} - {V_0} = {V_0}\left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T$
Now, the change in volume is given as,
$\vartriangle V = {V_0}\left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T - - - - - \left( 1 \right)$
Again we should find the change in density,
Let ${\rho _L}$ be the final density after change of temperature $\vartriangle T$,
${\rho _L} = \dfrac{{{\rho _{{L_0}}}}}{{\left( {1 + {\gamma _L}\vartriangle T} \right)}} - - - - - \left( 2 \right)$
Now, the mass collected can be represented as,
$\vartriangle m = \vartriangle V \times {\rho _{{L_{}}}}$
Substituting the value of $\vartriangle V$ and ${\rho _L}$ from equation $\left( 1 \right)$ and $\left( 2 \right)$ we get,
$\vartriangle m = {V_0}\left( {{\gamma _L} - {\gamma _C}} \right)\vartriangle T \times \dfrac{{{\rho _{{L_0}}}}}{{\left( {1 + {\gamma _L}\vartriangle T} \right)}}$
Interchanging the variables we get,
$\therefore \vartriangle T = \dfrac{{\vartriangle m}}{{{\rho _{{L_0}}}{V_0}({\gamma _L} - {\gamma _C}) - \vartriangle m{\gamma _L}}}$
So, the correct option is C.
Note: There is a possibility to overlook the volumetric expansion of the liquid inside the container. It is most important to recall that the volumetric expansion of solids helps to expand the metal surface which in turn allows less liquid to overflow. It must be subtracted while finding the mass overflow as done in the solution.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




