In a face centered cubic lattice, atom (A) occupies the corner positions and atom (B) occupies the face centre positions. If one atom of (B) is missing from one of the face centered points, the formula of the compound is
A.${A_2}{B_5}$
B.${A_2}B$
C.$A{B_2}$
D.${A_2}{B_3}$
Answer
552.5k+ views
Hint:We know that unit cell is the smallest repeating unit of the crystal lattice. We can call the building block of a crystal a unit cell. We have to know that a unit cell can be a face-centered cubic cell (FCC), body-centered cubic cell (BCC), and primitive cubic cell. We have to calculate the total number of atoms (A and B) per unit cell.
Complete step by step answer:We know that an FCC unit cell has atoms present at all the corners of the crystal lattice and atoms present at the centre of all the faces of the cube. Between 2 adjacent unit cells the atoms are shared at the face-centered and only ½ of each atom belongs to an individual cell.
In a face centered cubic unit cell, atoms are found in all corners of the lattice.
An atom is found at the center of every face.
Two adjacent unit-cells are shared by a face-center atom.
Twelve atoms per unit cell.
The coordination number of FCC is twelve and the total number of atoms per unit cell is four. The following diagram explains how the atoms are arranged in FCC which is given below as,
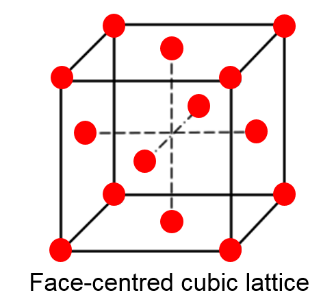
We know that in a face centered cubic lattice, atom (A) takes the corner positions. There are 8 corner positions and each position gives one eighth to the unit cell. So, we can total number of A atoms per unit cell as,
$\dfrac{1}{8} \times 8 = 1$
The total number of A atoms per unit cell is 1.
Atom B took the face centre positions. There are six face centre positions. One atom of B disappeared from one of the face centered points. So, there are 5 face centre positions which are occupied with B. Each such position gives one half to the unit cell. So, we total number of B atoms per unit cell as
$\dfrac{1}{2} \times 5 = 2.5$
The total number of B atoms per unit cell is 2.5.
So, the formula of the compound is $A{B_{2.5}}$ (or) ${A_2}{B_5}$.
Therefore, the option (A) is correct.
Note:As we know an example of a compound that has face-centered cubic lattice is sodium chloride. Lithium fluoride, lithium chloride, Sodium fluoride, potassium fluoride, potassium chloride etc are examples of compounds that contain face centered cubic structures. Simple cubic cell, and body centered cubic cell are the other two crystal structures.
Complete step by step answer:We know that an FCC unit cell has atoms present at all the corners of the crystal lattice and atoms present at the centre of all the faces of the cube. Between 2 adjacent unit cells the atoms are shared at the face-centered and only ½ of each atom belongs to an individual cell.
In a face centered cubic unit cell, atoms are found in all corners of the lattice.
An atom is found at the center of every face.
Two adjacent unit-cells are shared by a face-center atom.
Twelve atoms per unit cell.
The coordination number of FCC is twelve and the total number of atoms per unit cell is four. The following diagram explains how the atoms are arranged in FCC which is given below as,

We know that in a face centered cubic lattice, atom (A) takes the corner positions. There are 8 corner positions and each position gives one eighth to the unit cell. So, we can total number of A atoms per unit cell as,
$\dfrac{1}{8} \times 8 = 1$
The total number of A atoms per unit cell is 1.
Atom B took the face centre positions. There are six face centre positions. One atom of B disappeared from one of the face centered points. So, there are 5 face centre positions which are occupied with B. Each such position gives one half to the unit cell. So, we total number of B atoms per unit cell as
$\dfrac{1}{2} \times 5 = 2.5$
The total number of B atoms per unit cell is 2.5.
So, the formula of the compound is $A{B_{2.5}}$ (or) ${A_2}{B_5}$.
Therefore, the option (A) is correct.
Note:As we know an example of a compound that has face-centered cubic lattice is sodium chloride. Lithium fluoride, lithium chloride, Sodium fluoride, potassium fluoride, potassium chloride etc are examples of compounds that contain face centered cubic structures. Simple cubic cell, and body centered cubic cell are the other two crystal structures.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




