Identify the pair of alkanes which can exhibit chain isomerism and have a neo isomer respectively?
A. ${{\text{C}}_{3}}{{\text{H}}_{8}}\text{, }{{\text{C}}_{4}}{{\text{H}}_{10}}$
B. ${{\text{C}}_{4}}{{\text{H}}_{10}}\text{, }{{\text{C}}_{5}}{{\text{H}}_{12}}$
C. ${{\text{C}}_{5}}{{\text{H}}_{12}}\text{, }{{\text{C}}_{6}}{{\text{H}}_{14}}$
D. ${{\text{C}}_{6}}{{\text{H}}_{14}},{{\text{C}}_{7}}{{\text{H}}_{16}}$
Answer
558.6k+ views
Hint: The general configuration of alkanes is ${{\text{C}}_{\text{n}}}{{\text{H}}_{2\text{n}+\text{2}}}$, where n is the natural number. Draw the isomers of the given alkanes to check whether they show chain and neo isomerism or not. Also, the minimum requirement of carbon atoms for this isomerism should be known, which is 4 in chain and 5 in neo isomerism.
Complete answer:
Let us first discuss what chain and neo isomers are:
Chain isomers- The carbon compounds with the same molecular formula but have different atomic arrangements, the one with branches. The minimum number of carbon atoms required for chain isomerism are 4.
Neo isomers- In this, a carbon atom is attached to four other carbon atoms. Neo-isomers are the type of chain isomers only. The minimum number of carbon atoms required for neo isomers are 5.
Let us discuss the options one-by-one along with their chain and neo isomers:
A) ${{\text{C}}_{3}}{{\text{H}}_{8}}\text{, }{{\text{C}}_{4}}{{\text{H}}_{10}}$- The compound ${{\text{C}}_{3}}{{\text{H}}_{8}}$ is propane. As, the number of carbon atoms are less than 4, so it cannot have chain isomerism. ${{\text{C}}_{4}}{{\text{H}}_{10}}$ is butane. It has four carbon atoms, so it can form chain isomers. But, it cannot form neo isomers. The only isomer of butane is 2-methyl propane. The only isomer is
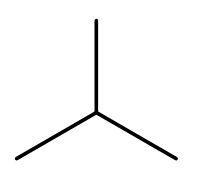
B) ${{\text{C}}_{4}}{{\text{H}}_{10}}\text{, }{{\text{C}}_{5}}{{\text{H}}_{12}}$- ${{\text{C}}_{4}}{{\text{H}}_{10}}$is butane. It has four carbon atoms, so it can form chain isomers. But, it cannot form neo isomers. As given in the above part the only isomer of butane is 2-methyl propane.
The compound ${{\text{C}}_{5}}{{\text{H}}_{12}}$ is pentane. It has both chain and neo-isomer, as it has 5 carbon atoms. The chain isomer is 2-methyl butane. The neo isomer is 2,2-dimethylpropane.
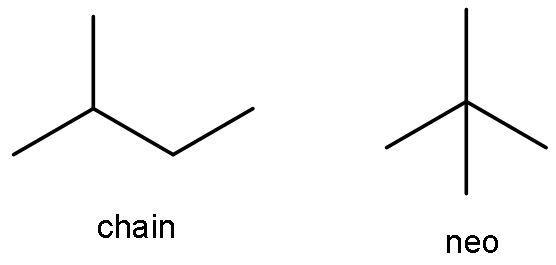
C)${{\text{C}}_{5}}{{\text{H}}_{12}}\text{, }{{\text{C}}_{6}}{{\text{H}}_{14}}$: As discussed in the above solution part, the compound ${{\text{C}}_{5}}{{\text{H}}_{12}}$ is pentane. It has both chain and neo-isomer. The chain isomer is 2-methyl butane. The neo isomer is 2, 2-dimethylpropane.
The compound ${{\text{C}}_{6}}{{\text{H}}_{14}}$ is hexane. It has both chain and neo-isomer, as it has more than 5 carbon atoms. The chain isomers are 2-methyl pentane and 2,3-dimethyl butane. The neo isomers are 2,2-dimethylbutane.
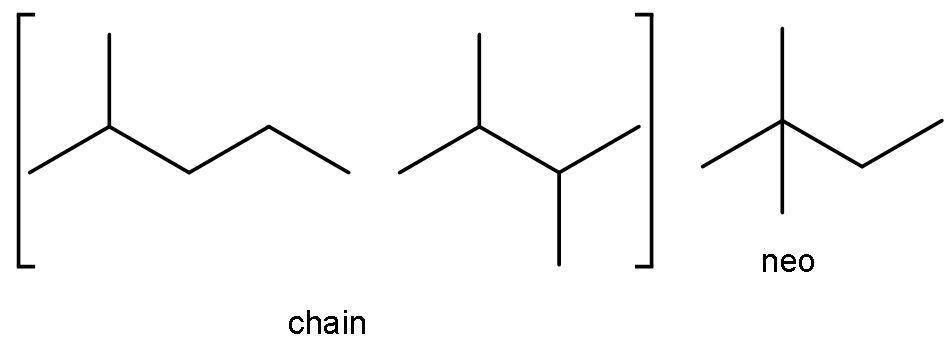
D)${{\text{C}}_{6}}{{\text{H}}_{14}},{{\text{C}}_{7}}{{\text{H}}_{16}}$: As given in the above solution part we know that hexane has both chain and neo-isomers. The chain isomers are 2-methyl pentane and 2,3-dimethyl butane. The neo isomers are 2, 2-dimethylbutane.
The compound ${{\text{C}}_{7}}{{\text{H}}_{16}}$ is heptane. It has both chain and neo-isomer, as it has more than 5 carbon atoms. The chain isomers are 2-methyl hexane, 2,3-dimethyl pentane and 2,4-dimethyl pentane.
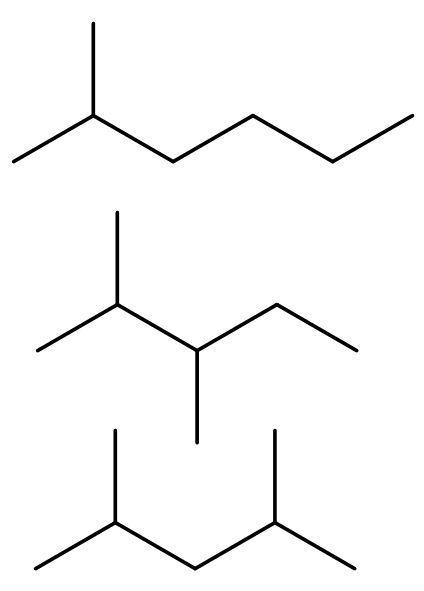
The neo isomers are 2,2-dimethyl-3-methyl butane, 2,2-dimethyl pentane and 3,3-dimethyl pentane.
${{\text{C}}_{5}}{{\text{H}}_{12}}\text{, }{{\text{C}}_{6}}{{\text{H}}_{14}}$ and ${{\text{C}}_{6}}{{\text{H}}_{14}},{{\text{C}}_{7}}{{\text{H}}_{16}}$ are the pair of alkanes which can exhibit chain isomerism and has a neo isomer respectively.
The correct options are (c) and (d).
Note:
As the branching in the carbon chain increases, the surface area decreases and vander waal forces of attraction decrease. The force of attraction decreases, thus, the boiling point of alkane is also decreased. The neo isomers have less boiling point than chain isomers.
Complete answer:
Let us first discuss what chain and neo isomers are:
Chain isomers- The carbon compounds with the same molecular formula but have different atomic arrangements, the one with branches. The minimum number of carbon atoms required for chain isomerism are 4.
Neo isomers- In this, a carbon atom is attached to four other carbon atoms. Neo-isomers are the type of chain isomers only. The minimum number of carbon atoms required for neo isomers are 5.
Let us discuss the options one-by-one along with their chain and neo isomers:
A) ${{\text{C}}_{3}}{{\text{H}}_{8}}\text{, }{{\text{C}}_{4}}{{\text{H}}_{10}}$- The compound ${{\text{C}}_{3}}{{\text{H}}_{8}}$ is propane. As, the number of carbon atoms are less than 4, so it cannot have chain isomerism. ${{\text{C}}_{4}}{{\text{H}}_{10}}$ is butane. It has four carbon atoms, so it can form chain isomers. But, it cannot form neo isomers. The only isomer of butane is 2-methyl propane. The only isomer is

B) ${{\text{C}}_{4}}{{\text{H}}_{10}}\text{, }{{\text{C}}_{5}}{{\text{H}}_{12}}$- ${{\text{C}}_{4}}{{\text{H}}_{10}}$is butane. It has four carbon atoms, so it can form chain isomers. But, it cannot form neo isomers. As given in the above part the only isomer of butane is 2-methyl propane.
The compound ${{\text{C}}_{5}}{{\text{H}}_{12}}$ is pentane. It has both chain and neo-isomer, as it has 5 carbon atoms. The chain isomer is 2-methyl butane. The neo isomer is 2,2-dimethylpropane.

C)${{\text{C}}_{5}}{{\text{H}}_{12}}\text{, }{{\text{C}}_{6}}{{\text{H}}_{14}}$: As discussed in the above solution part, the compound ${{\text{C}}_{5}}{{\text{H}}_{12}}$ is pentane. It has both chain and neo-isomer. The chain isomer is 2-methyl butane. The neo isomer is 2, 2-dimethylpropane.
The compound ${{\text{C}}_{6}}{{\text{H}}_{14}}$ is hexane. It has both chain and neo-isomer, as it has more than 5 carbon atoms. The chain isomers are 2-methyl pentane and 2,3-dimethyl butane. The neo isomers are 2,2-dimethylbutane.

D)${{\text{C}}_{6}}{{\text{H}}_{14}},{{\text{C}}_{7}}{{\text{H}}_{16}}$: As given in the above solution part we know that hexane has both chain and neo-isomers. The chain isomers are 2-methyl pentane and 2,3-dimethyl butane. The neo isomers are 2, 2-dimethylbutane.
The compound ${{\text{C}}_{7}}{{\text{H}}_{16}}$ is heptane. It has both chain and neo-isomer, as it has more than 5 carbon atoms. The chain isomers are 2-methyl hexane, 2,3-dimethyl pentane and 2,4-dimethyl pentane.

The neo isomers are 2,2-dimethyl-3-methyl butane, 2,2-dimethyl pentane and 3,3-dimethyl pentane.
${{\text{C}}_{5}}{{\text{H}}_{12}}\text{, }{{\text{C}}_{6}}{{\text{H}}_{14}}$ and ${{\text{C}}_{6}}{{\text{H}}_{14}},{{\text{C}}_{7}}{{\text{H}}_{16}}$ are the pair of alkanes which can exhibit chain isomerism and has a neo isomer respectively.
The correct options are (c) and (d).
Note:
As the branching in the carbon chain increases, the surface area decreases and vander waal forces of attraction decrease. The force of attraction decreases, thus, the boiling point of alkane is also decreased. The neo isomers have less boiling point than chain isomers.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




