
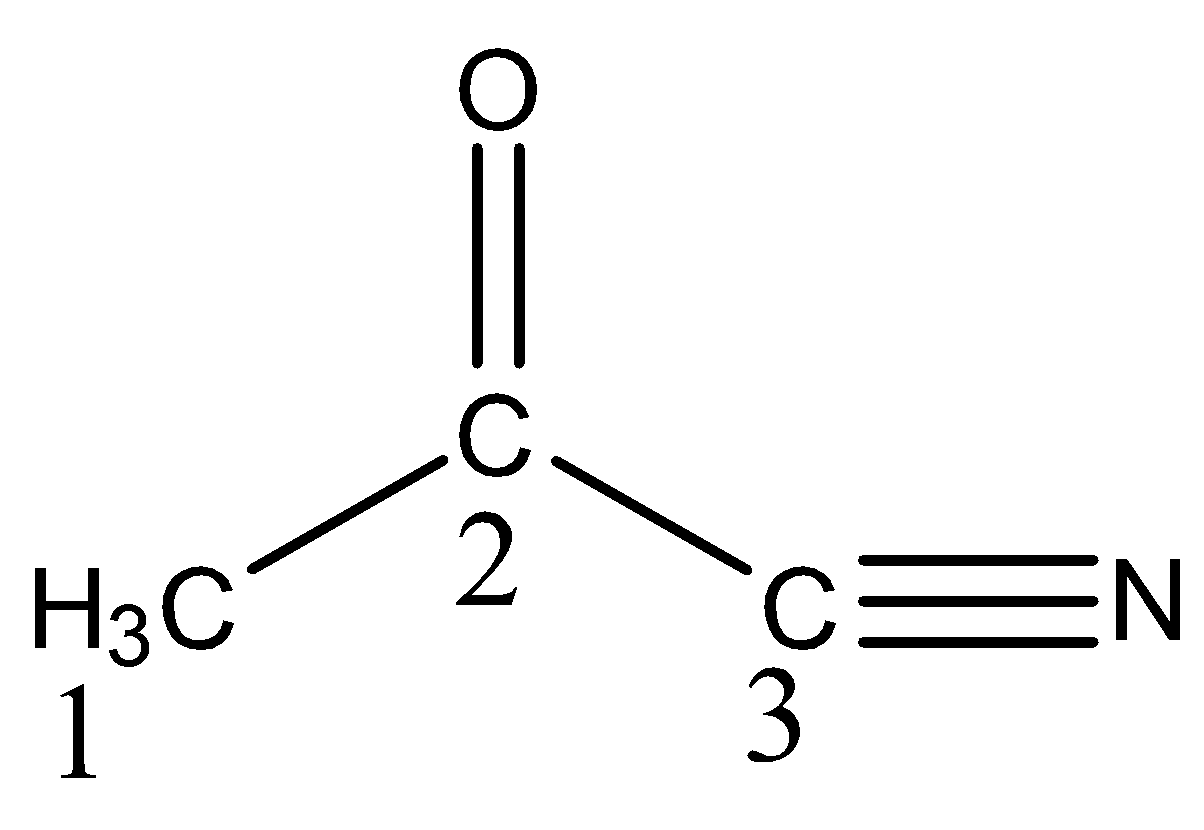
What hybrid orbitals are employed by carbon atom ${{1, 2 and 3}}$respectively as labelled in the compound shown?
(A) ${{s}}{{{p}}^{{3}}}{{, sp , sp}}$
(B) ${{s}}{{{p}}^{{2}}}{{ , s}}{{{p}}^{{3}}}{{ , sp}}$
(C) ${{s}}{{{p}}^{{3}}}{{ , s}}{{{p}}^{{2}}}{{ , sp}}$
(D) ${{s}}{{{p}}^{{3}}}{{ , s}}{{{p}}^{{2}}}{{ , s}}{{{p}}^{{2}}}$

Answer
580.8k+ views
Hint: First we will see the number of bonds between the carbon and its substituents and see the type of overlapping of the orbitals involved in the formation of the compound. Every hybridized orbital has a different shape than the pure orbital.
Complete step by step answer:
First, we will see the type of overlapping and shape of the hybridized orbitals.
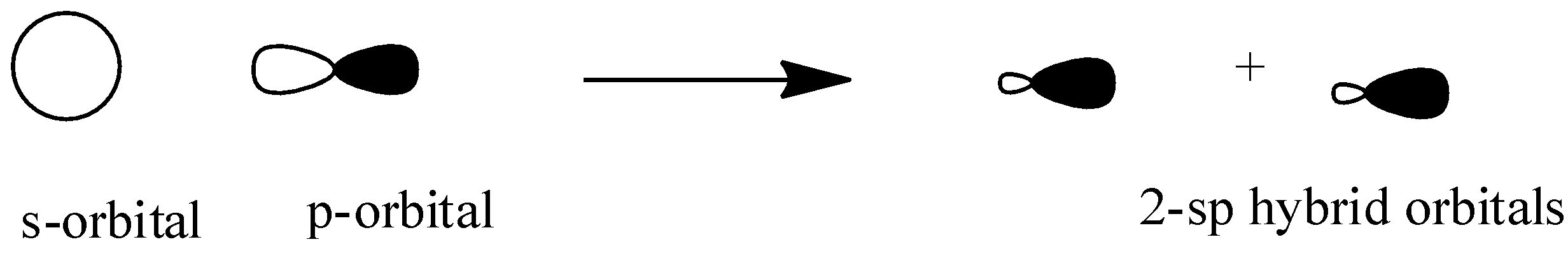
\[{{sp}}\]: The \[{{sp}}\] hybridization occurs when one s-orbital and one p-orbital overlap each other head on to form two \[{{sp}}\] hybridized orbitals having linear shape. Here two p-orbitals are unhybridized so they form ${{2\pi }}$ bonds.
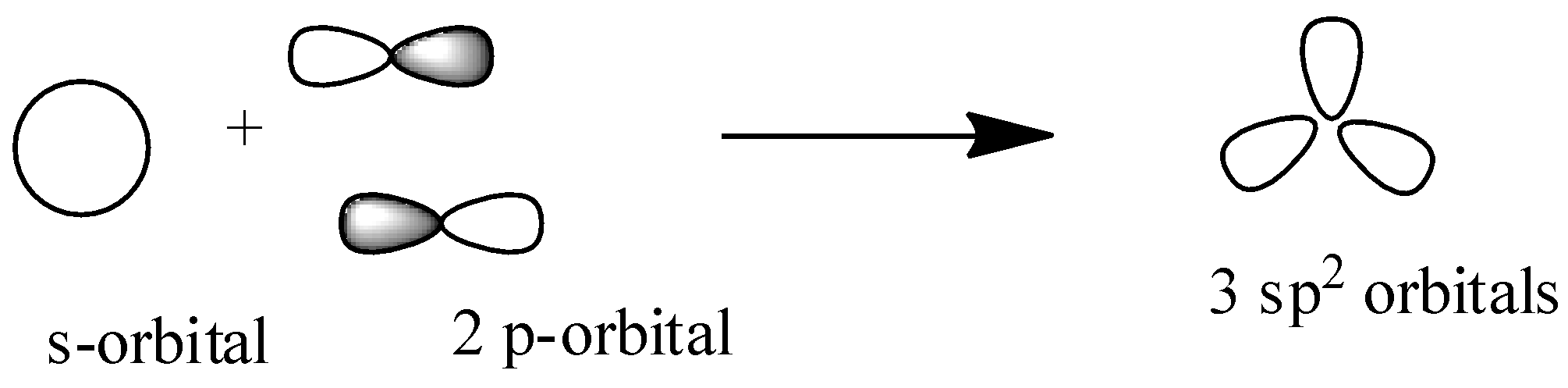
${{s}}{{{p}}^{{2}}}$: Here in this hybridization one s-orbital and two p-orbitals overlap to form ${{3 s}}{{{p}}^{{2}}}$ hybridized orbitals and here one p-orbital is unhybridized so that it can form ${{\pi }}$-bond between two carbon or between carbon and a heteroatom. So ${{s}}{{{p}}^{{2}}}$ hybridized orbital has ${{3\sigma orbitals and 1\pi orbital}}$ to make single and double bond. It has trigonal shape
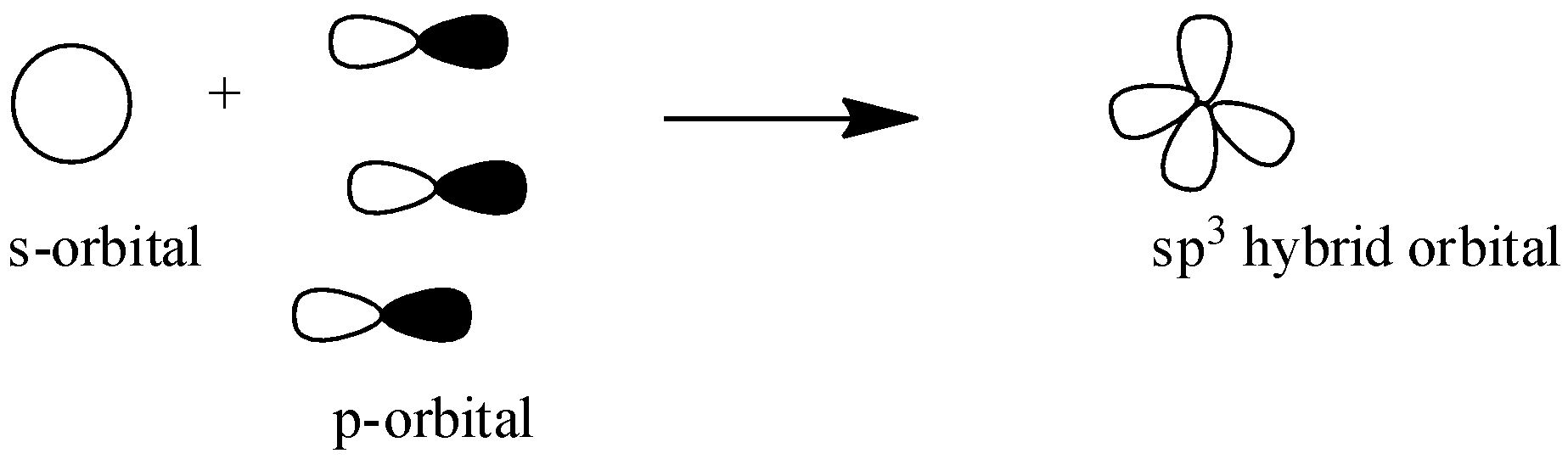
${{s}}{{{p}}^{{3}}}$ : In this hybridization one s-orbital overlaps with three p-orbital to form three ${{s}}{{{p}}^{{3}}}$ orbitals. Here we don’t have any unhybridized p-orbital to form ${{ \pi }}$ bond only ${{4\sigma }}$ bonds are formed and structure of these hybridized orbitals are tetrahedron.
So now in structure carbon ${{1}}$ has ${{4\sigma\; bonds\; no \;\pi \; bond}}$ so the hybridization of carbon 1 is $sp^3$
When we see carbon ${{2}}$ we observe it has ${{3\sigma \; and \; 1\pi \;bond}}$ and has trigonal planar structure so this fits the condition for ${{s}}{{{p}}^{{2}}}$ hybridization.
Now last carbon ${{3}}$ we see it has ${{2\sigma \; and \;2\pi \; bonds}}$ in it and structure is also linear then the hybridization is sp
So, the correct answer is Option C .
Note: By knowing the hybridization of the carbon or any element in a molecule we can know the nature of bond and characteristic of it whether the bond is strong or weak or it makes the molecule electron donating or withdrawing group.
The triple bond is the strongest among the single , double and triple bond because single bond only have ${{1\sigma }}$bond and double bond has ${{1\sigma \; and\; 1\pi\; bond}}$ whereas triple bond have ${{1\sigma \; and\; 2\pi \;\ bonds}}$ in it which makes it strongest.
Complete step by step answer:
First, we will see the type of overlapping and shape of the hybridized orbitals.
\[{{sp}}\]: The \[{{sp}}\] hybridization occurs when one s-orbital and one p-orbital overlap each other head on to form two \[{{sp}}\] hybridized orbitals having linear shape. Here two p-orbitals are unhybridized so they form ${{2\pi }}$ bonds.

${{s}}{{{p}}^{{2}}}$: Here in this hybridization one s-orbital and two p-orbitals overlap to form ${{3 s}}{{{p}}^{{2}}}$ hybridized orbitals and here one p-orbital is unhybridized so that it can form ${{\pi }}$-bond between two carbon or between carbon and a heteroatom. So ${{s}}{{{p}}^{{2}}}$ hybridized orbital has ${{3\sigma orbitals and 1\pi orbital}}$ to make single and double bond. It has trigonal shape

${{s}}{{{p}}^{{3}}}$ : In this hybridization one s-orbital overlaps with three p-orbital to form three ${{s}}{{{p}}^{{3}}}$ orbitals. Here we don’t have any unhybridized p-orbital to form ${{ \pi }}$ bond only ${{4\sigma }}$ bonds are formed and structure of these hybridized orbitals are tetrahedron.

So now in structure carbon ${{1}}$ has ${{4\sigma\; bonds\; no \;\pi \; bond}}$ so the hybridization of carbon 1 is $sp^3$
When we see carbon ${{2}}$ we observe it has ${{3\sigma \; and \; 1\pi \;bond}}$ and has trigonal planar structure so this fits the condition for ${{s}}{{{p}}^{{2}}}$ hybridization.
Now last carbon ${{3}}$ we see it has ${{2\sigma \; and \;2\pi \; bonds}}$ in it and structure is also linear then the hybridization is sp
So, the correct answer is Option C .
Note: By knowing the hybridization of the carbon or any element in a molecule we can know the nature of bond and characteristic of it whether the bond is strong or weak or it makes the molecule electron donating or withdrawing group.
The triple bond is the strongest among the single , double and triple bond because single bond only have ${{1\sigma }}$bond and double bond has ${{1\sigma \; and\; 1\pi\; bond}}$ whereas triple bond have ${{1\sigma \; and\; 2\pi \;\ bonds}}$ in it which makes it strongest.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Can anyone list 10 advantages and disadvantages of friction




