\[{{H}_{2}}O\] is liquid but \[{{H}_{2}}S\] is gas. This is because of:
(A)- larger size of S
(B)- acidic nature of \[{{H}_{2}}S\]
(C)- hydrogen bonding
(D)- small size of O
Answer
629.4k+ views
Hint: Hydrogen bonding is a type of weak interaction between a hydrogen atom bonded to an atom of high electron affinity (e.g. N, O, and F) and an electron rich atom with high electron affinity. It is, therefore, a weak bond between an electronegative atom and a hydrogen atom bonded to another electronegative atom.
Complete step by step solution:
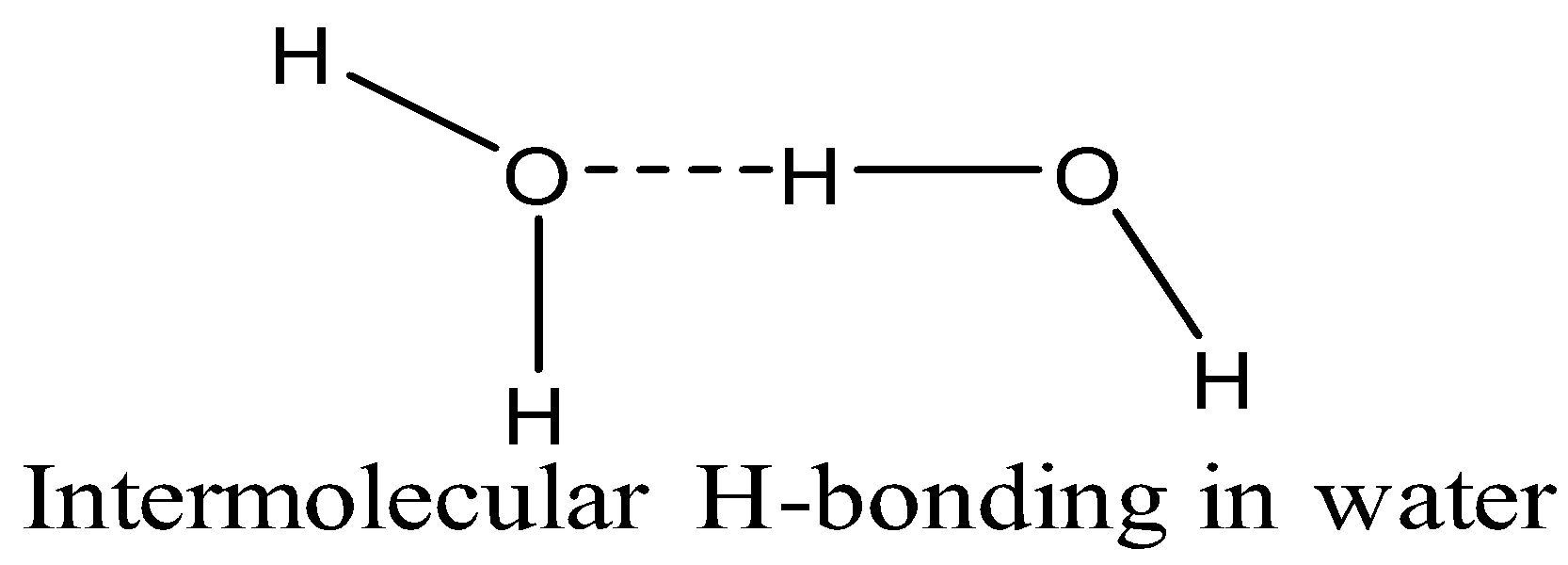
\[{{H}_{2}}O\] and \[{{H}_{2}}S\] are the hydrides of group 16. The hydride of oxygen i.e. \[{{H}_{2}}O\] is liquid whereas the hydride of sulphur is in gaseous state. The reason for this difference in their physical states is the ability of water molecules to undergo intermolecular hydrogen bonding.
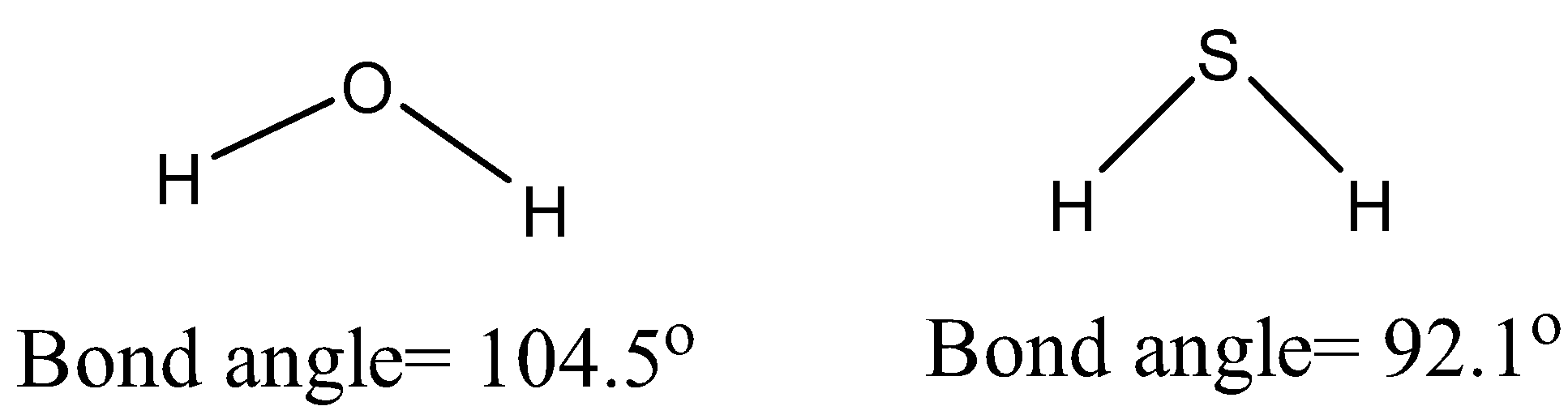
Oxygen is the most electronegative of all the elements in group 16. Since it has more electron affinity than sulphur, its hydride i.e. \[{{H}_{2}}O\], undergoes intermolecular hydrogen bonding. O of one \[{{H}_{2}}O\] molecule is bonded with the hydrogen of another \[{{H}_{2}}O\]molecule. In this way, \[{{H}_{2}}O\] molecules are held together by H-bonding. No such interactions are present among \[{{H}_{2}}S\] molecules and are only held together by weak van der Waals forces of attraction.
As electronegativity decreases with the increase in sizes down the group, other elements including S, Se, and Te do not form hydrogen bonds. Therefore, their hydrides are all in gaseous state.
 The correct option is (C).
The correct option is (C).
Additional Information: The hydrides of other elements of group 16 excluding \[{{H}_{2}}O\] are poisonous, unpleasant and foul smelling gases. They all are volatile and \[{{H}_{2}}O\] has the highest boiling point.
Note: Due to the smaller size of O, its electronegativity is higher than S. But intermolecular hydrogen bonding is the most adequate explanation for the difference in physical states of \[{{H}_{2}}O\] and\[{{H}_{2}}S\].
Complete step by step solution:
\[{{H}_{2}}O\] and \[{{H}_{2}}S\] are the hydrides of group 16. The hydride of oxygen i.e. \[{{H}_{2}}O\] is liquid whereas the hydride of sulphur is in gaseous state. The reason for this difference in their physical states is the ability of water molecules to undergo intermolecular hydrogen bonding.
Oxygen is the most electronegative of all the elements in group 16. Since it has more electron affinity than sulphur, its hydride i.e. \[{{H}_{2}}O\], undergoes intermolecular hydrogen bonding. O of one \[{{H}_{2}}O\] molecule is bonded with the hydrogen of another \[{{H}_{2}}O\]molecule. In this way, \[{{H}_{2}}O\] molecules are held together by H-bonding. No such interactions are present among \[{{H}_{2}}S\] molecules and are only held together by weak van der Waals forces of attraction.
As electronegativity decreases with the increase in sizes down the group, other elements including S, Se, and Te do not form hydrogen bonds. Therefore, their hydrides are all in gaseous state.


Additional Information: The hydrides of other elements of group 16 excluding \[{{H}_{2}}O\] are poisonous, unpleasant and foul smelling gases. They all are volatile and \[{{H}_{2}}O\] has the highest boiling point.
Note: Due to the smaller size of O, its electronegativity is higher than S. But intermolecular hydrogen bonding is the most adequate explanation for the difference in physical states of \[{{H}_{2}}O\] and\[{{H}_{2}}S\].
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




