Graphite is
This question has multiple correct options
A. A good conductor
B. $s{p^2}$ hybridised
C. An amorphous solid
D. A covalent crystal
Answer
610.5k+ views
Hint: It is referred to as plumbago, it is a crystalline form of the element carbon with its atom arranged in hexagonal structure.
Complete step by step answer:
Graphite is composed of flat two dimensional sheets of carbon atoms. Each sheet is a hexagonal net of C atoms, and may be regarded as a fused system of benzene rings. The layers are held together by relatively weaker van der waals force.
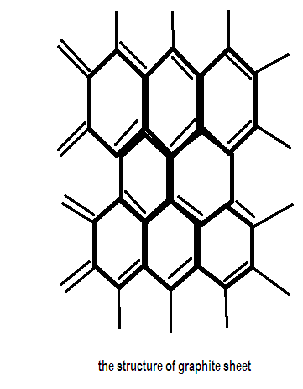
Graphite and diamonds are the only two naturally formed polymers of carbon. Graphite is essentially a two dimensional, planar crystal structure. Soft,greasy dark grayish coloured crystalline solid.
Carbon in graphite has a $s{p^2}$ type of hybridization and is linked to three other carbon atoms in a hexagonal planar structure. Since only three of the valence electrons of each carbon atom are involved the remaining unhybridized p orbital of each carbon atom having an electron overlaps with its counterparts of the adjacent carbon atoms in the same layer to form pi-bonds. Hence the pi-electrons are delocalized in the whole sheet which is why Graphite is an excellent conductor of heat and electricity and has the highest natural strength and stiffness of any material and is very resistant to chemical attack.
At the same time, it is one of the lightest of all reinforcing agents and has high natural lubricity. Graphite is a covalent crystal and crystalline. It is quite hard because of the strong covalent bonding throughout the lattice.
Hence the correct option are A,B and D.
Note:
It occurs naturally in hexagonal structure under high pressures and temperatures it converts to diamond. Graphite is used in pencils and lubricants. It is a good conductor of heat and electricity. Its high conductivity makes it useful in electronic products such as electrodes, batteries, and solar panels.
Complete step by step answer:
Graphite is composed of flat two dimensional sheets of carbon atoms. Each sheet is a hexagonal net of C atoms, and may be regarded as a fused system of benzene rings. The layers are held together by relatively weaker van der waals force.

Graphite and diamonds are the only two naturally formed polymers of carbon. Graphite is essentially a two dimensional, planar crystal structure. Soft,greasy dark grayish coloured crystalline solid.
Carbon in graphite has a $s{p^2}$ type of hybridization and is linked to three other carbon atoms in a hexagonal planar structure. Since only three of the valence electrons of each carbon atom are involved the remaining unhybridized p orbital of each carbon atom having an electron overlaps with its counterparts of the adjacent carbon atoms in the same layer to form pi-bonds. Hence the pi-electrons are delocalized in the whole sheet which is why Graphite is an excellent conductor of heat and electricity and has the highest natural strength and stiffness of any material and is very resistant to chemical attack.
At the same time, it is one of the lightest of all reinforcing agents and has high natural lubricity. Graphite is a covalent crystal and crystalline. It is quite hard because of the strong covalent bonding throughout the lattice.
Hence the correct option are A,B and D.
Note:
It occurs naturally in hexagonal structure under high pressures and temperatures it converts to diamond. Graphite is used in pencils and lubricants. It is a good conductor of heat and electricity. Its high conductivity makes it useful in electronic products such as electrodes, batteries, and solar panels.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




