
Given, $CH\equiv C-C{{H}_{3}}\xrightarrow[1%\text{ }HgS{{O}_{4}}]{40%\text{ }{{H}_{2}}S{{O}_{4}}}B$
Then, B is:
(a)- Acetone
(b)- Trichloroacetone
(c)- Acetaldehyde
(d)- Chloral
Answer
531.6k+ views
Hint: The given compound on the reactant side is propan-1-yne and it is a member of the alkyne group. When the alkynes react with 40% sulfuric acid and 1% of mercuric sulfate, then the hydration reaction occurs, i.e., there will be the addition of water molecules to the compound forming alcohol then, there will be tautomerism in the compound.
Complete answer:
The given compound on the reactant side is propan-1-yne and it is a member of the alkyne group because there are three carbon atoms in the chain and at the first carbon there is a triple bond.
When the alkynes react with 40% sulfuric acid and 1% of mercuric sulfate, then the hydration reaction occurs, i.e., there will be the addition of water molecules to the compound forming alcohol then, there will be tautomerism in the compound.
So, when the propyne reacts with 40% sulfuric acid and 1% of mercuric sulfate, then a hydrogen atom of the water gets added to the first carbon atom and the hydroxyl ion of water gets added to the second carbon atom forming alkene alcohol. Now, there will be tautomerism, i.e., the hydrogen atom will shift to the third position. So, the hydrogen atom of the hydroxyl group will move towards the $C{{H}_{2}}$ group. The reaction is given below:
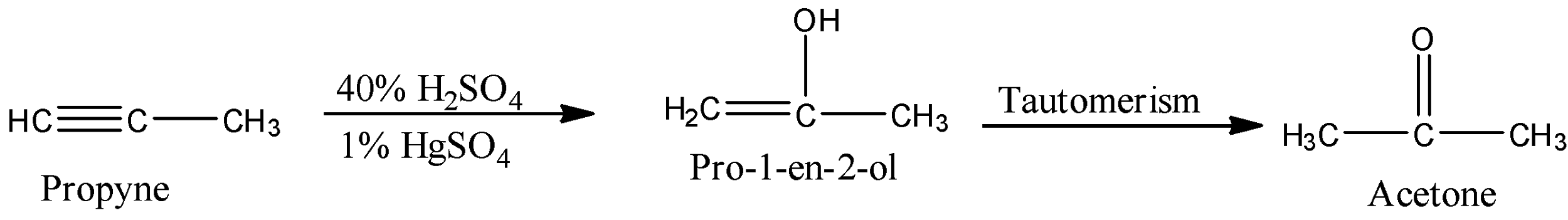
So, the product formed in the reaction is acetone.
Therefore, the correct answer is an option (a)- Acetone
Note:
The addition of water molecules to the alkyne takes place through Markovnikov’s rule, i.e., the negative part of the water molecule (hydroxyl ion) gets attached to the carbon atom having a lesser number of hydrogen atoms.
Complete answer:
The given compound on the reactant side is propan-1-yne and it is a member of the alkyne group because there are three carbon atoms in the chain and at the first carbon there is a triple bond.
When the alkynes react with 40% sulfuric acid and 1% of mercuric sulfate, then the hydration reaction occurs, i.e., there will be the addition of water molecules to the compound forming alcohol then, there will be tautomerism in the compound.
So, when the propyne reacts with 40% sulfuric acid and 1% of mercuric sulfate, then a hydrogen atom of the water gets added to the first carbon atom and the hydroxyl ion of water gets added to the second carbon atom forming alkene alcohol. Now, there will be tautomerism, i.e., the hydrogen atom will shift to the third position. So, the hydrogen atom of the hydroxyl group will move towards the $C{{H}_{2}}$ group. The reaction is given below:

So, the product formed in the reaction is acetone.
Therefore, the correct answer is an option (a)- Acetone
Note:
The addition of water molecules to the alkyne takes place through Markovnikov’s rule, i.e., the negative part of the water molecule (hydroxyl ion) gets attached to the carbon atom having a lesser number of hydrogen atoms.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




