For $0 \cdot 90{\text{ gm}}$of an organic compound ${C_4}{H_{10}}{O_2}$, ${\text{A}}$, when treated with Sodium gives $224{\text{ ml}}$ of hydrogen at ${\text{NTP}}$. Compound $A$ can be separated into fractions ${\text{B and C}}$ by crystallisation of which the fraction ${\text{B}}$ is resolved into optical isomers ${\text{D and E}}$. Write down the structural formula of ${\text{A and E}}$ with proper reasoning.
Answer
587.4k+ views
Hint:Compound ${\text{A}}$ must be containing active hydrogens to give ${H_2}$ when treated with ${\text{Na}}$. It should be symmetrical as it can be fractionated into two. Optical isomers are enantiomers. Hence, ${\text{D and E}}$ will be the mirror image of each other.
Complete step by step answer:
Step (1):
The given compound is ${C_4}{H_{10}}{O_2}$. Its molecular mass will be:
$\left( {4 \times {\text{Mass of Carbon + 10}} \times {\text{ Mass of Hydrogen + 2}} \times {\text{Mass of Oxygen}}} \right)$
= $\left( {4 \times 12 + 10 \times 1 + 2 \times 16} \right)$
= $90{\text{ gm mo}}{{\text{l}}^{ - 1}}$
Step (2):
Given mass of compound is $0 \cdot 90{\text{ gm}}$.
So, Number of moles of the compound = $\dfrac{{{\text{Given mass}}}}{{{\text{Molar mass}}}}$
${\text{ = }}\dfrac{{0 \cdot 90}}{{90}}$
= $0 \cdot 01{\text{ mol}}$
So, Number of moles of the compound is $0 \cdot 01{\text{ mol}}$.
Step (3):
It is given that $224{\text{ ml}}$ of ${H_2}$ is released. Molar volume of ${H_2}$ at ${\text{NTP}}$ is $22 \cdot 4{\text{ L}}$.
So, Number of Moles of ${H_2}$ = $\dfrac{{{\text{Given Volume}}}}{{{\text{Molar volume}}}}$
= $\dfrac{{224{\text{ ml}}}}{{22400{\text{ ml}}}}$
$ = 0 \cdot 01{\text{ mol}}$
Since, $1{\text{ mole}}$ of ${H_2}$ contains ${\text{2 moles of H}}$. So, $0 \cdot 01{\text{ mol}}$ of ${H_2}$ contains $0 \cdot 02{\text{ moles of H}}$.
Step (4):
As the compound contains active hydrogen to undergo reaction with Sodium, it must contain some $\left( {{\text{ - OH}}} \right)$ group to provide those active hydrogens. The reaction will be:
$2{C_4}{H_{10}}{O_2} + 2Na \to 2{C_4}{H_9}{O_2}Na + {H_2}$
As there are $0 \cdot 02{\text{ moles of active H}}$ present in $0 \cdot 01{\text{ mol}}$ of compound. So, the compound must contain ${\text{2 ( - OH) }}$ groups. Hence, the structural formula of $A$ will be ${C_4}{H_8}{\left( {OH} \right)_2}$.
Following are the possible structures of ${\text{A}}$:
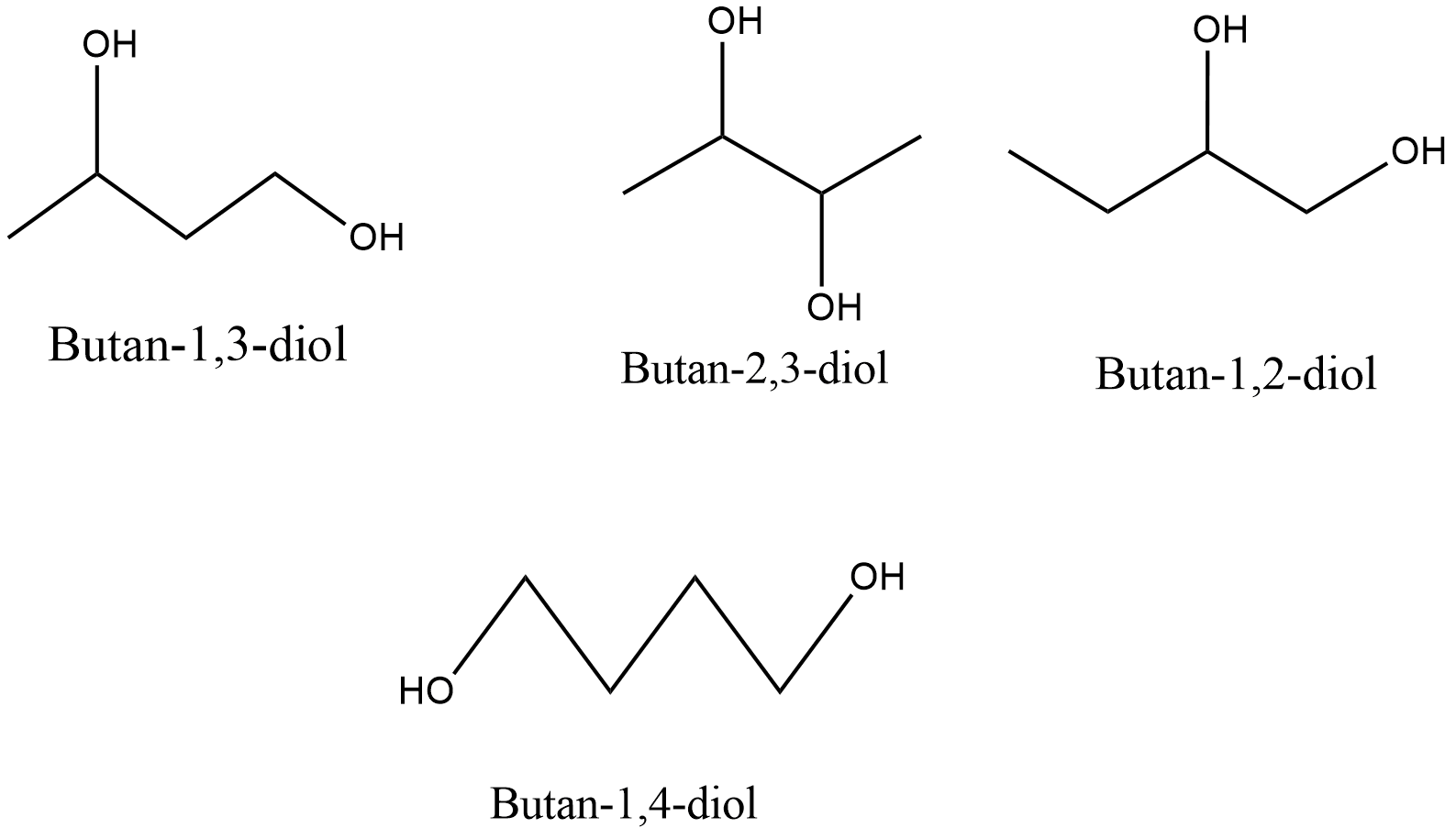
Among above all only ${\text{Butan - 2,3 - diol}}$ can be possible because for rest, Fisher projections cannot be drawn to see the symmetry for further answering the question which says, ${\text{A}}$ can be fractionated into
${\text{B and C}}$ whose structures will be:
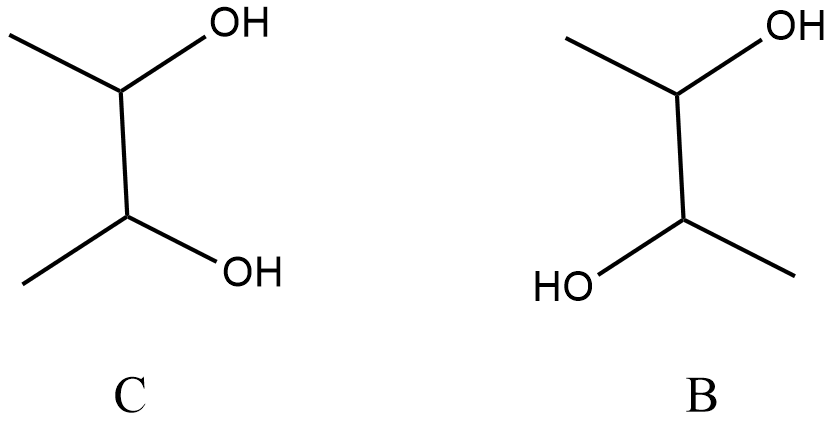
Step (5):
It is given that ${\text{B}}$ can be resolved into two optical isomers ${\text{D and E}}$, and optical isomers are enantiomers which are mirror images of each other. That means B should not be a meso compound, so ${\text{C}}$ is taken as a meso compound. The structure of ${\text{D and E}}$ will be:
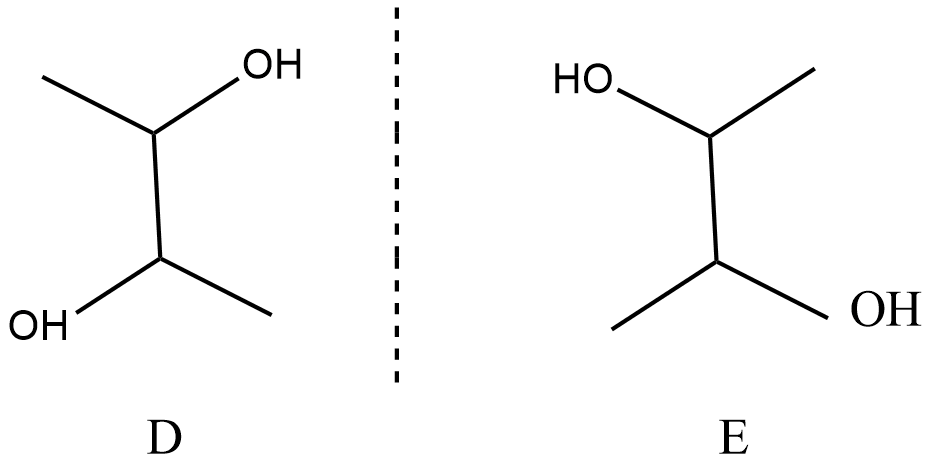
Hence, the Structural formula for ${\text{A and E}}$ is the same as ${C_4}{H_8}{\left( {OH} \right)_2}$ .
Note:
For optically active compounds, it should have the same type of ligand on the same side of the molecule, as in case of ${\text{C}}$ here, which is a meso compound or repeated pair of ligands. Plane of symmetry is a sign of optically inactive compounds.
Complete step by step answer:
Step (1):
The given compound is ${C_4}{H_{10}}{O_2}$. Its molecular mass will be:
$\left( {4 \times {\text{Mass of Carbon + 10}} \times {\text{ Mass of Hydrogen + 2}} \times {\text{Mass of Oxygen}}} \right)$
= $\left( {4 \times 12 + 10 \times 1 + 2 \times 16} \right)$
= $90{\text{ gm mo}}{{\text{l}}^{ - 1}}$
Step (2):
Given mass of compound is $0 \cdot 90{\text{ gm}}$.
So, Number of moles of the compound = $\dfrac{{{\text{Given mass}}}}{{{\text{Molar mass}}}}$
${\text{ = }}\dfrac{{0 \cdot 90}}{{90}}$
= $0 \cdot 01{\text{ mol}}$
So, Number of moles of the compound is $0 \cdot 01{\text{ mol}}$.
Step (3):
It is given that $224{\text{ ml}}$ of ${H_2}$ is released. Molar volume of ${H_2}$ at ${\text{NTP}}$ is $22 \cdot 4{\text{ L}}$.
So, Number of Moles of ${H_2}$ = $\dfrac{{{\text{Given Volume}}}}{{{\text{Molar volume}}}}$
= $\dfrac{{224{\text{ ml}}}}{{22400{\text{ ml}}}}$
$ = 0 \cdot 01{\text{ mol}}$
Since, $1{\text{ mole}}$ of ${H_2}$ contains ${\text{2 moles of H}}$. So, $0 \cdot 01{\text{ mol}}$ of ${H_2}$ contains $0 \cdot 02{\text{ moles of H}}$.
Step (4):
As the compound contains active hydrogen to undergo reaction with Sodium, it must contain some $\left( {{\text{ - OH}}} \right)$ group to provide those active hydrogens. The reaction will be:
$2{C_4}{H_{10}}{O_2} + 2Na \to 2{C_4}{H_9}{O_2}Na + {H_2}$
As there are $0 \cdot 02{\text{ moles of active H}}$ present in $0 \cdot 01{\text{ mol}}$ of compound. So, the compound must contain ${\text{2 ( - OH) }}$ groups. Hence, the structural formula of $A$ will be ${C_4}{H_8}{\left( {OH} \right)_2}$.
Following are the possible structures of ${\text{A}}$:

Among above all only ${\text{Butan - 2,3 - diol}}$ can be possible because for rest, Fisher projections cannot be drawn to see the symmetry for further answering the question which says, ${\text{A}}$ can be fractionated into
${\text{B and C}}$ whose structures will be:

Step (5):
It is given that ${\text{B}}$ can be resolved into two optical isomers ${\text{D and E}}$, and optical isomers are enantiomers which are mirror images of each other. That means B should not be a meso compound, so ${\text{C}}$ is taken as a meso compound. The structure of ${\text{D and E}}$ will be:

Hence, the Structural formula for ${\text{A and E}}$ is the same as ${C_4}{H_8}{\left( {OH} \right)_2}$ .
Note:
For optically active compounds, it should have the same type of ligand on the same side of the molecule, as in case of ${\text{C}}$ here, which is a meso compound or repeated pair of ligands. Plane of symmetry is a sign of optically inactive compounds.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




