
Find the number of lone pairs of electrons on $Xe$ in $XeO{F_4}$.
Answer
591k+ views
Hint: To find the number of lone pairs of electrons on $Xe$, we need to draw the structure of $XeO{F_4}$. We should also be aware of the oxidation states in which the central atom $Xe$ can exist, so as to know the number of electrons involved in bonding and number of lone pairs.
Complete step by step answer:
Xenon is found in multiple oxidation states such as $0$, $ + 2$, $ + 4$, $ + 6$ and $ + 8$. Due to this it forms a number of oxides, some of these oxides are stable while some are not.
Xenon also forms oxide fluorides which consists of both oxygen and fluorine atoms bonded to $Xe$. One of such oxide fluorides is xenon oxytetrafluoride, i.e., $XeO{F_4}$.
The O-atom is in $ - 2$ oxidation state and each F-atom is in $ - 1$ oxidation state. The overall charge on the molecule is zero, so the oxidation state of $Xe$ is,
$x + ( - 2) + ( - 1 \times 4) = 0$
$ \Rightarrow x - 2 - 4 = 0$
$ \Rightarrow x - 6 = 0$
$ \Rightarrow x = + 6$
$Xe$ is a noble gas and it has $8$ valence electrons. As it is in $ + 6$ oxidation state, it means it has shared $6$ electrons with the bonded atoms. Therefore, only $2$ more electrons are left in a non-bonded state. That is, one lone pair of electrons is present on $Xe$.
Structure of $XeO{F_4}$:
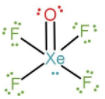
Note:
We can also find the number of lone pairs directly by calculating the number of electrons shared by $Xe$ to the bonded atoms, without the need to calculate the oxidation number. As $Xe$ has $8$ valence electrons, it is bonded to O-atom with a double bond, so $2$ electrons are shared; $4$ F-atoms are singly bonded to $Xe$, so $4$ electrons are shared with fluorine. The rest $2$ electrons are present as lone pairs of electrons.
Complete step by step answer:
Xenon is found in multiple oxidation states such as $0$, $ + 2$, $ + 4$, $ + 6$ and $ + 8$. Due to this it forms a number of oxides, some of these oxides are stable while some are not.
Xenon also forms oxide fluorides which consists of both oxygen and fluorine atoms bonded to $Xe$. One of such oxide fluorides is xenon oxytetrafluoride, i.e., $XeO{F_4}$.
The O-atom is in $ - 2$ oxidation state and each F-atom is in $ - 1$ oxidation state. The overall charge on the molecule is zero, so the oxidation state of $Xe$ is,
$x + ( - 2) + ( - 1 \times 4) = 0$
$ \Rightarrow x - 2 - 4 = 0$
$ \Rightarrow x - 6 = 0$
$ \Rightarrow x = + 6$
$Xe$ is a noble gas and it has $8$ valence electrons. As it is in $ + 6$ oxidation state, it means it has shared $6$ electrons with the bonded atoms. Therefore, only $2$ more electrons are left in a non-bonded state. That is, one lone pair of electrons is present on $Xe$.
Structure of $XeO{F_4}$:

Note:
We can also find the number of lone pairs directly by calculating the number of electrons shared by $Xe$ to the bonded atoms, without the need to calculate the oxidation number. As $Xe$ has $8$ valence electrons, it is bonded to O-atom with a double bond, so $2$ electrons are shared; $4$ F-atoms are singly bonded to $Xe$, so $4$ electrons are shared with fluorine. The rest $2$ electrons are present as lone pairs of electrons.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




