$EDT{{A}^{4-}}$ is ethylene diamine tetraacetate ion. The total number of $-N-Co-O$ bond angles in ${{[Co(EDTA)]}^{-}}$ complex ion is:
Answer
609.9k+ views
Hint: To get the total number of bond angles, first try to draw the chemical structure of the ethylene diamine tetraacetate ion and then see what structure it gives when it is treated with cobalt atoms. Draw the chemical structure of the complex formed, then you will get an idea about the number of bond angles present in the complex.
Complete step by step answer:
So, before proceeding to find the answer, let us know about ethylene diamine tetraacetate, in short referred to as EDTA.
So, EDTA is a hexadentate ligand, which implies that it binds multiple (six) times. It binds twice at the nitrogen and four at the oxygen atoms. It has an atomic structure a lot like a claw. The chemical structure of ethylene diamine tetraacetate ion i.e. $EDT{{A}^{4-}}$ is shown in the figure below:
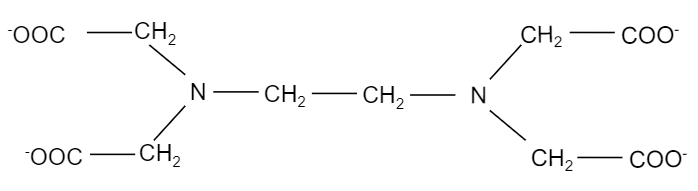
When ethylene diamine tetraacetate ion is treated with cobalt element, it forms a complex, ${{[Co(EDTA)]}^{-}}$ where, cobalt binds with two nitrogen atoms and four oxygen ions present in the EDTA structure. The structure of the complex is shown below:
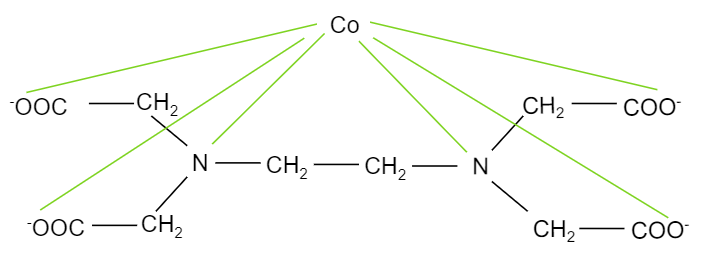
The oxygen atoms having negative charges and the two nitrogen atoms will be donating electrons forming bonds with cobalt. So, we can see that, in the complex, each nitrogen has four $-N-Co-O$ bond angles. And there are two nitrogen atoms bonded with cobalt. So, the total number of $-N-Co-O$ bond angles will be eight.
Hence, the total number of $-N-Co-O$ bond angles in ${{[Co(EDTA)]}^{-}}$ complex ion is eight.
Note: Metal analysis can be done by using the process of titration of the ethylene diamine tetraacetate and a metal where metal ion acts as the indicator. One of the most famous and most common uses of EDTA is to determine the hardness of the water.
Complete step by step answer:
So, before proceeding to find the answer, let us know about ethylene diamine tetraacetate, in short referred to as EDTA.
So, EDTA is a hexadentate ligand, which implies that it binds multiple (six) times. It binds twice at the nitrogen and four at the oxygen atoms. It has an atomic structure a lot like a claw. The chemical structure of ethylene diamine tetraacetate ion i.e. $EDT{{A}^{4-}}$ is shown in the figure below:
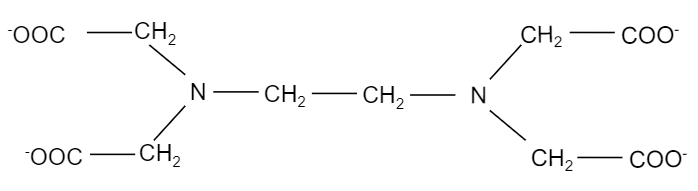
When ethylene diamine tetraacetate ion is treated with cobalt element, it forms a complex, ${{[Co(EDTA)]}^{-}}$ where, cobalt binds with two nitrogen atoms and four oxygen ions present in the EDTA structure. The structure of the complex is shown below:

The oxygen atoms having negative charges and the two nitrogen atoms will be donating electrons forming bonds with cobalt. So, we can see that, in the complex, each nitrogen has four $-N-Co-O$ bond angles. And there are two nitrogen atoms bonded with cobalt. So, the total number of $-N-Co-O$ bond angles will be eight.
Hence, the total number of $-N-Co-O$ bond angles in ${{[Co(EDTA)]}^{-}}$ complex ion is eight.
Note: Metal analysis can be done by using the process of titration of the ethylene diamine tetraacetate and a metal where metal ion acts as the indicator. One of the most famous and most common uses of EDTA is to determine the hardness of the water.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




