Draw the structure of 3-methylpent-2-ene.
Answer
604.8k+ views
Hint:We are given an IUPAC name of the compound. The method of naming organic compounds is known as the IUPAC nomenclature. In the given name, suffix –ene is written thus, a double bond must be present.
Complete answer
We are given an IUPAC name of the compound i.e. 3-methylpent-2-ene.
pent- means that the parent alkane is pentane. Pentane means there are five carbon atoms present.
3-methyl means there is a methyl $\left( { - {\text{C}}{{\text{H}}_3}} \right)$ group attached to the third carbon of the parent alkane. pent-2-ene means there is a double bond to the second carbon.
Thus, the structure of 3-methylpent-2-ene is as follows:
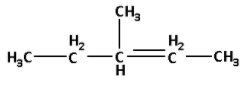
In the structure, we can see that there are five carbon atoms, the methyl group is attached to the third carbon and the double bond is attached to the second carbon.
Note:The rules for writing IUPAC name of alkenes are as follows:
i) Select the longest continuous chain of carbon atoms containing carbon-carbon double bonds in the structure of the molecule.
ii) Number the carbon atoms in the selected carbon chain from the end which is nearest to the double bond.
iii) Count the number of carbon atoms in the chain. This is the parent alkane.
iv) The name of alkene is written by replacing suffix ‘ane’ in the parent alkane by ‘ene’
v) Write the number indicating the position of the double position before prefix ‘ene’.
vi) Assign a number to each substituent according to the carbon atom it is attached. If there are two substituents on the same carbon, assign the same number to them.
Complete answer
We are given an IUPAC name of the compound i.e. 3-methylpent-2-ene.
pent- means that the parent alkane is pentane. Pentane means there are five carbon atoms present.
3-methyl means there is a methyl $\left( { - {\text{C}}{{\text{H}}_3}} \right)$ group attached to the third carbon of the parent alkane. pent-2-ene means there is a double bond to the second carbon.
Thus, the structure of 3-methylpent-2-ene is as follows:

In the structure, we can see that there are five carbon atoms, the methyl group is attached to the third carbon and the double bond is attached to the second carbon.
Note:The rules for writing IUPAC name of alkenes are as follows:
i) Select the longest continuous chain of carbon atoms containing carbon-carbon double bonds in the structure of the molecule.
ii) Number the carbon atoms in the selected carbon chain from the end which is nearest to the double bond.
iii) Count the number of carbon atoms in the chain. This is the parent alkane.
iv) The name of alkene is written by replacing suffix ‘ane’ in the parent alkane by ‘ene’
v) Write the number indicating the position of the double position before prefix ‘ene’.
vi) Assign a number to each substituent according to the carbon atom it is attached. If there are two substituents on the same carbon, assign the same number to them.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




