How do you draw a Newman projection for hexane while sighting down $ {C_2} - {C_3} $ ?
Answer
508.8k+ views
Hint: For the given problem, we first need to figure out the normal structure of hexane which is a straight chain of six carbon atoms with all valances satisfied by the hydrogen atoms. Then figuring out the $ {C_2} - {C_3} $ and then fixing them in Newman projection template. After fixing the groups on the template, we have the Newman structure of hexane.
Complete Step By Step Answer:
1. Draw the structure of hexane.
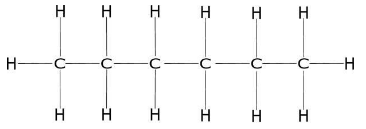
2. Convert the drawn structure from $ {C_2} $ and $ {C_3} $ to a wedge-dash structure.
3. Identifies the $ {C_2} $ and $ {C_3} $ groups.
The main chain of hexane is the horizontal zigzag line of carbon atoms. $ {C_1} $ is on the left.
The groups of $ {C_2} $ are $ H,H\& C{H_3} $ . At $ {C_3} $ are $ H,H,\& C{H_2}C{H_2}C{H_3} $ .
4. Draw the Newman projection template.
5. Attach the group to the carbon in the template.
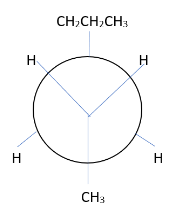
Shows the numerator from the left. The group of $ {C_2} $ is on the previous carbon atom. Place the $ C{H_3} $ group at the bottom. The two hydrogen atoms move to the other bond. The group of $ {C_3} $ moves to the rear carbon. The bulky $ C{H_2}C{H_2}C{H_3} $ group is at the top and two hydrogen atoms are on the other bond.
This is the most stable conformer because it has displaced volumetric methyl and propyl groups.
Note:
The Newman projection, useful in alkane stereochemistry, visualizes the structure of a chemical bond from front to back, with the front atom represented as a point and the rear carbon as a circle. The carbon atom in front is called near, while the atom behind is called far.
Complete Step By Step Answer:
1. Draw the structure of hexane.

2. Convert the drawn structure from $ {C_2} $ and $ {C_3} $ to a wedge-dash structure.
3. Identifies the $ {C_2} $ and $ {C_3} $ groups.
The main chain of hexane is the horizontal zigzag line of carbon atoms. $ {C_1} $ is on the left.
The groups of $ {C_2} $ are $ H,H\& C{H_3} $ . At $ {C_3} $ are $ H,H,\& C{H_2}C{H_2}C{H_3} $ .
4. Draw the Newman projection template.
5. Attach the group to the carbon in the template.

Shows the numerator from the left. The group of $ {C_2} $ is on the previous carbon atom. Place the $ C{H_3} $ group at the bottom. The two hydrogen atoms move to the other bond. The group of $ {C_3} $ moves to the rear carbon. The bulky $ C{H_2}C{H_2}C{H_3} $ group is at the top and two hydrogen atoms are on the other bond.
This is the most stable conformer because it has displaced volumetric methyl and propyl groups.
Note:
The Newman projection, useful in alkane stereochemistry, visualizes the structure of a chemical bond from front to back, with the front atom represented as a point and the rear carbon as a circle. The carbon atom in front is called near, while the atom behind is called far.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




