How does phenol differ from benzene?
Answer
582.6k+ views
Hint: The phenol and benzene have both structural differences at only one carbon where the alcohol group is attached. One can elaborate on the differences in both the chemicals about the physical and chemical properties.
Complete step by step answer:
1) First of all, as we know both phenols and benzene are aromatic structures and they differ from each other in the attachment of a group of alcohol to one carbon. Now let us see each chemical structure in detail as below,
2) Phenol:
i) Structure of phenol:
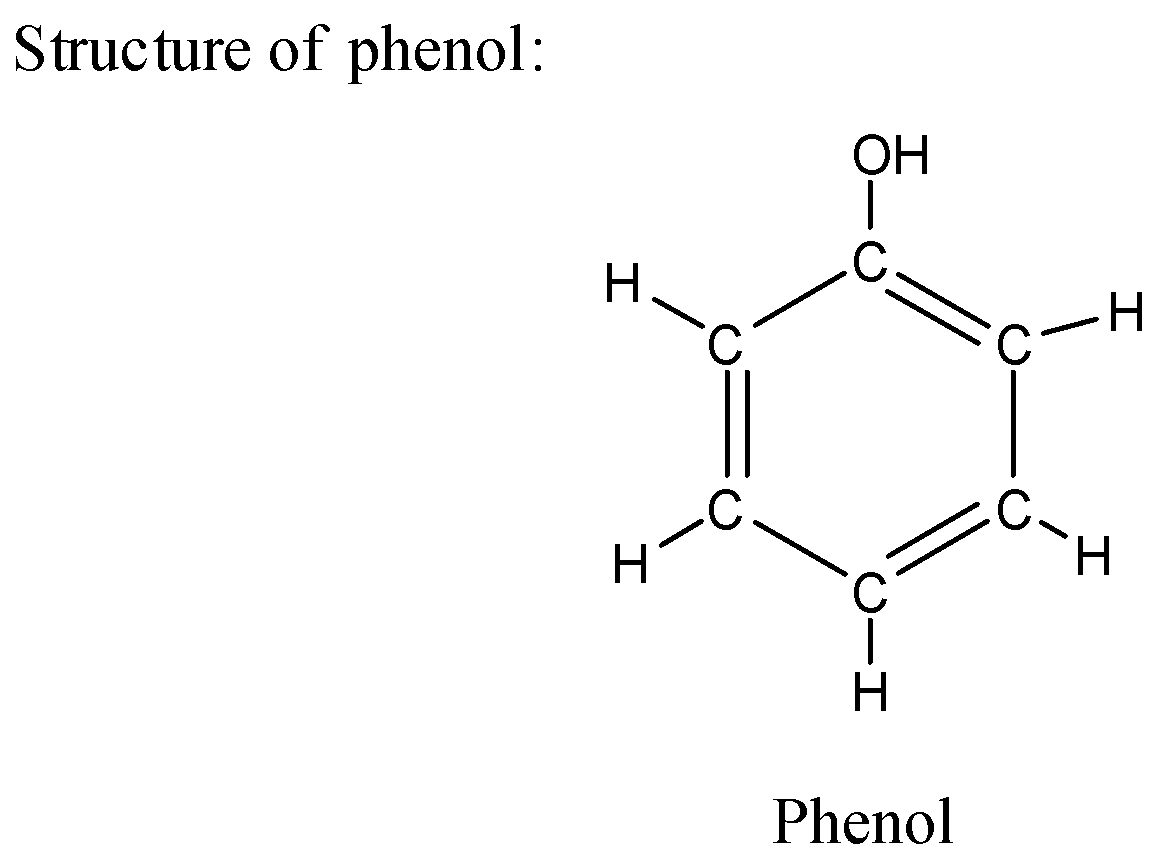
ii) The phenol has a molecular formula as ${C_6}{H_6}OH$ which has an alcoholic functional group in its structure.
iii) The phenol is present in solid form and has a white crystalline appearance.
iv) Phenol has a very strong odor which is very unique and can be detected in the lab easily by smell.
v) Phenol is acidic in nature which means it donates proton easily.
vi) Phenol is easily soluble in solvents like water.
3) Benzene:
i) Structure of benzene:
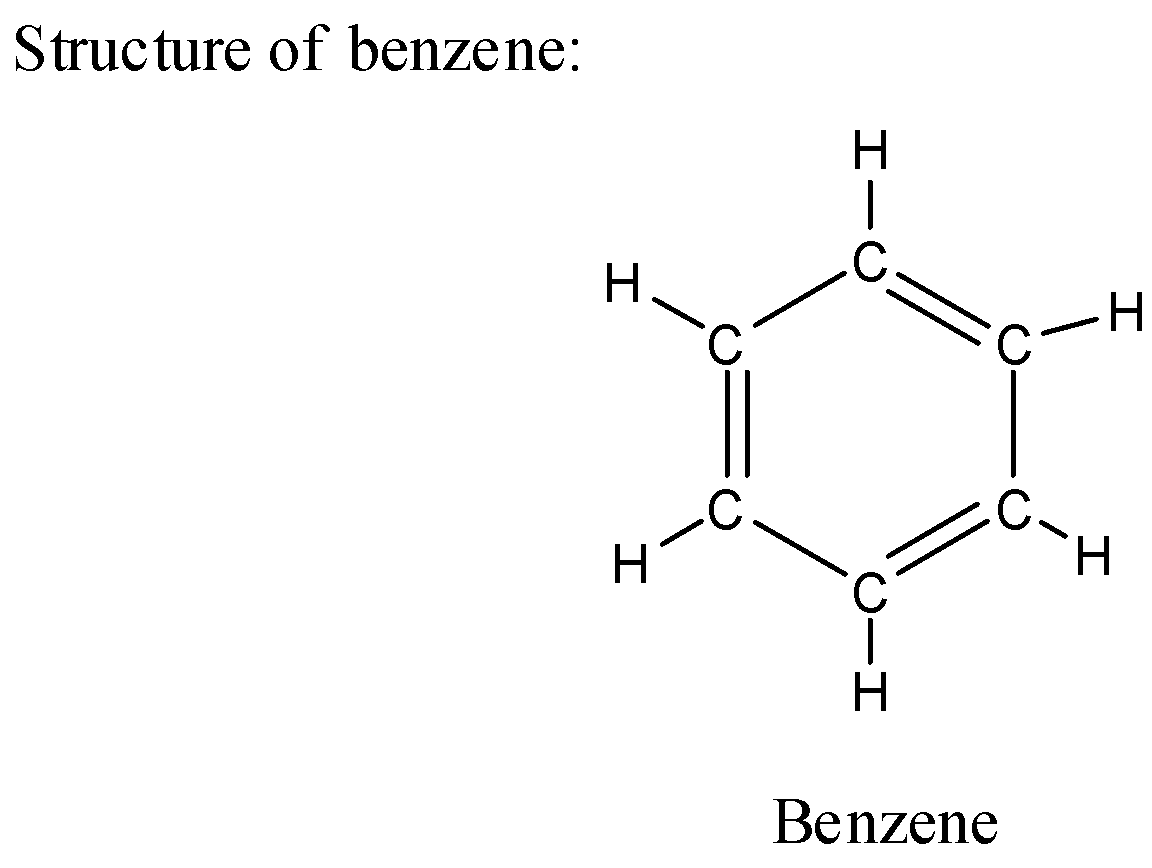
ii) The chemical structure of benzene has a molecular formula as ${C_6}{H_6}$ where each carbon in the ring has one hydrogen atom.
iii) The benzene is present in liquid form and is colorless in appearance.
iv) Benzene has a very sweet odor which is unique from other odors.
v) The benzene has neutral nature in chemical reactivity and hence it is mostly used as a solvent in many chemical reactions.
vi) The benzene is less soluble in water as compared to phenol which is more soluble in water.
Note:
The phenol is acidic in nature despite having an alcoholic group in its structure. The hydrogen atom donation forms a phenoxide ion which is stable due to aromaticity and that makes phenol acidic. Both the phenol and benzene are volatile where benzene has a faster rate of evaporation.
Complete step by step answer:
1) First of all, as we know both phenols and benzene are aromatic structures and they differ from each other in the attachment of a group of alcohol to one carbon. Now let us see each chemical structure in detail as below,
2) Phenol:
i) Structure of phenol:

ii) The phenol has a molecular formula as ${C_6}{H_6}OH$ which has an alcoholic functional group in its structure.
iii) The phenol is present in solid form and has a white crystalline appearance.
iv) Phenol has a very strong odor which is very unique and can be detected in the lab easily by smell.
v) Phenol is acidic in nature which means it donates proton easily.
vi) Phenol is easily soluble in solvents like water.
3) Benzene:
i) Structure of benzene:

ii) The chemical structure of benzene has a molecular formula as ${C_6}{H_6}$ where each carbon in the ring has one hydrogen atom.
iii) The benzene is present in liquid form and is colorless in appearance.
iv) Benzene has a very sweet odor which is unique from other odors.
v) The benzene has neutral nature in chemical reactivity and hence it is mostly used as a solvent in many chemical reactions.
vi) The benzene is less soluble in water as compared to phenol which is more soluble in water.
Note:
The phenol is acidic in nature despite having an alcoholic group in its structure. The hydrogen atom donation forms a phenoxide ion which is stable due to aromaticity and that makes phenol acidic. Both the phenol and benzene are volatile where benzene has a faster rate of evaporation.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE




