Dimethyl ether and ethyl alcohol are:
A.metamers
B.homologues
C.functional isomers
D.position isomers
Answer
590.4k+ views
Hint: We know that isomerism is the phenomenon in which two or more compounds have the same chemical formula but different structures. The compounds showing isomerism are known as isomers. There are different types of isomers, such as, position isomers, tautomers, chain isomers etc.
Complete step by step solution:
Let’s first discuss metamers, homologues, functional isomers and position isomers in detail.
Metamers are the isomers which differ with respect to the nature of alkyl groups around a particular functional group. Thus, they belong to the same family. For example, the molecular formula ${{\rm{C}}_{\rm{4}}}{{\rm{H}}_{{\rm{10}}}}{\rm{O}}$ possesses three metamers.
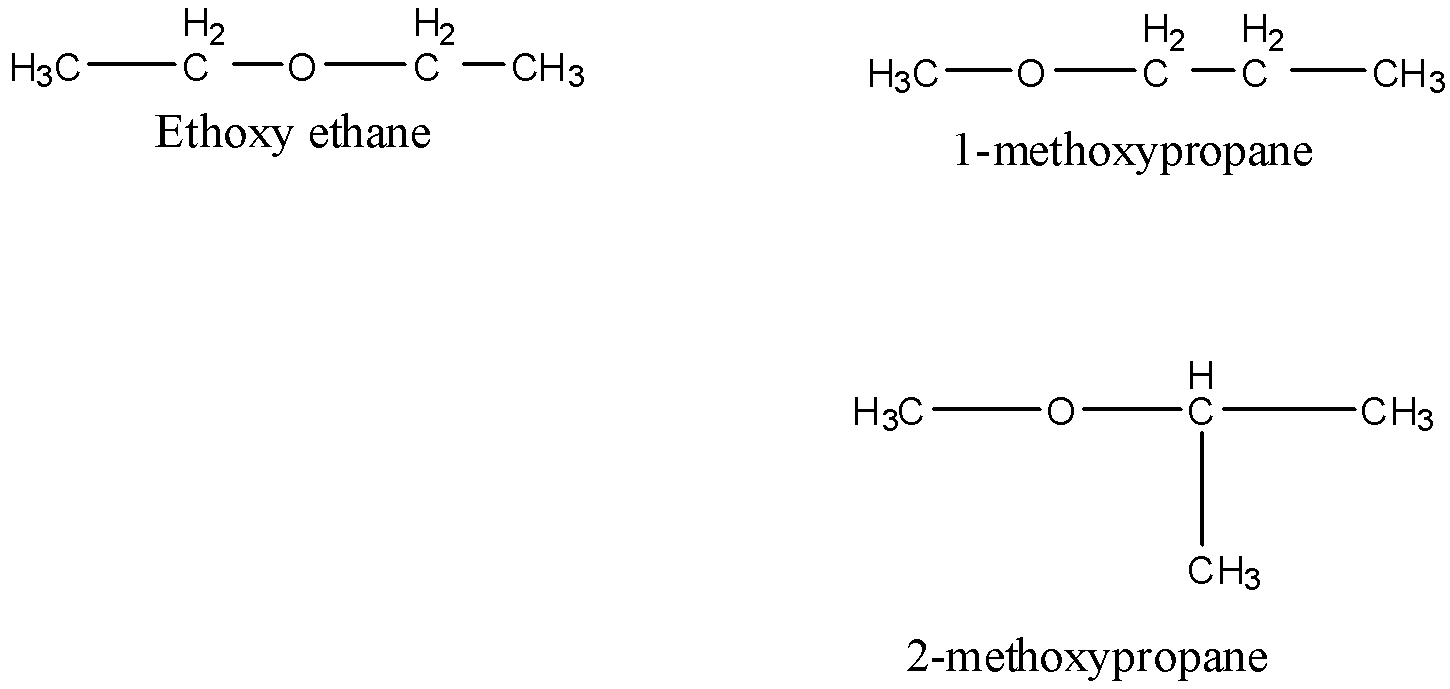
All the isomers belong to the family of ether.
Now, we discuss homologues. Homologues are series of compounds represented by the same molecular formula and a difference of${\rm{C}}{{\rm{H}}_{\rm{2}}}$ group is observed between two successive homologues. Such as, ${\rm{C}}{{\rm{H}}_{\rm{4}}}$ and ${{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{6}}}$.
Functional isomers are the isomers that differ with respect to the nature of functional groups. So, both the isomers belong to different families. For example, ${{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{6}}}{\rm{O}}$ has two functional isomers.

Position isomers are the isomers which differ with respect to the position of multiple bonds (double or triple), substituents or functional groups. For example, ${{\rm{C}}_{\rm{3}}}{{\rm{H}}_{\rm{7}}}{\rm{OH}}$ has two position isomers.
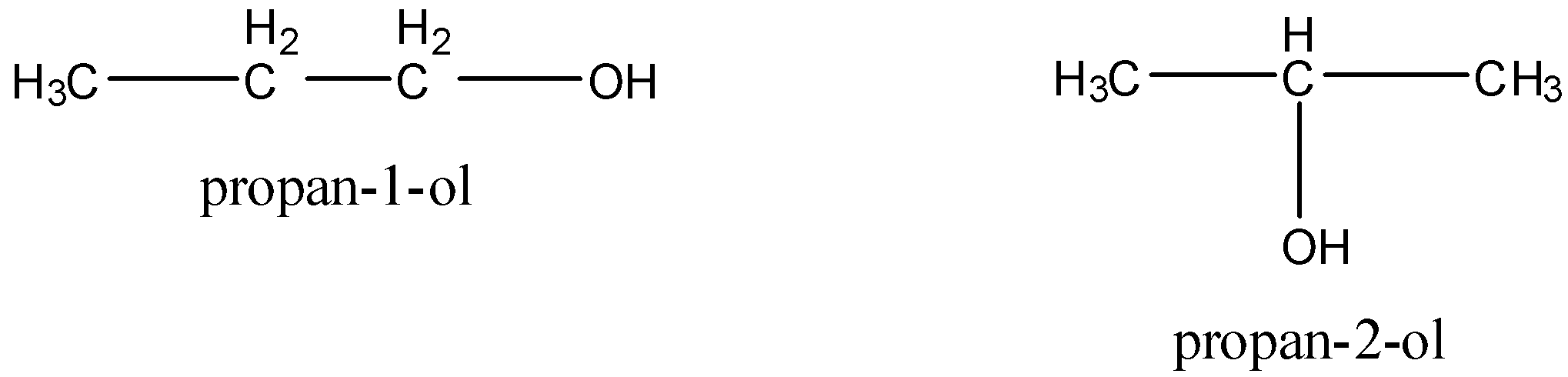
Now, we draw the structure of dimethyl ether and ethyl alcohol.

So, both the compounds differ by their functional groups. So, they are functional isomers.
Therefore, the correct answer is Option C.
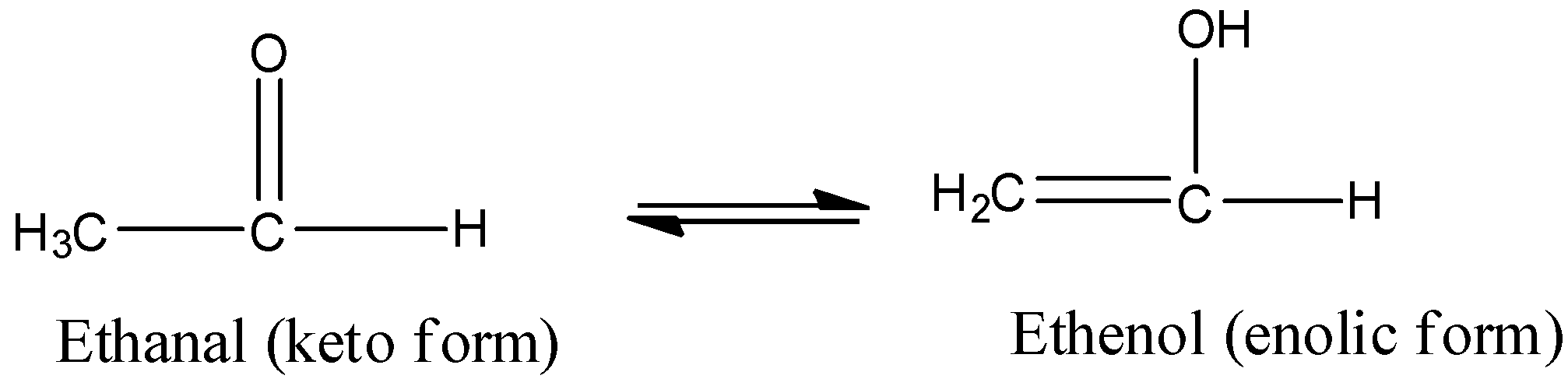
Note: Tautomers are actually functional isomers which exist simultaneously and also in dynamic equilibrium. The isomerism in this case is termed as tautomerism. It is of different types but the most common among them is the keto-enol tautomerism. This isomerism arises due to 1,3 migration of the hydrogen atom from carbon to oxygen atom and vice versa. For example,
Complete step by step solution:
Let’s first discuss metamers, homologues, functional isomers and position isomers in detail.
Metamers are the isomers which differ with respect to the nature of alkyl groups around a particular functional group. Thus, they belong to the same family. For example, the molecular formula ${{\rm{C}}_{\rm{4}}}{{\rm{H}}_{{\rm{10}}}}{\rm{O}}$ possesses three metamers.

All the isomers belong to the family of ether.
Now, we discuss homologues. Homologues are series of compounds represented by the same molecular formula and a difference of${\rm{C}}{{\rm{H}}_{\rm{2}}}$ group is observed between two successive homologues. Such as, ${\rm{C}}{{\rm{H}}_{\rm{4}}}$ and ${{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{6}}}$.
Functional isomers are the isomers that differ with respect to the nature of functional groups. So, both the isomers belong to different families. For example, ${{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{6}}}{\rm{O}}$ has two functional isomers.

Position isomers are the isomers which differ with respect to the position of multiple bonds (double or triple), substituents or functional groups. For example, ${{\rm{C}}_{\rm{3}}}{{\rm{H}}_{\rm{7}}}{\rm{OH}}$ has two position isomers.
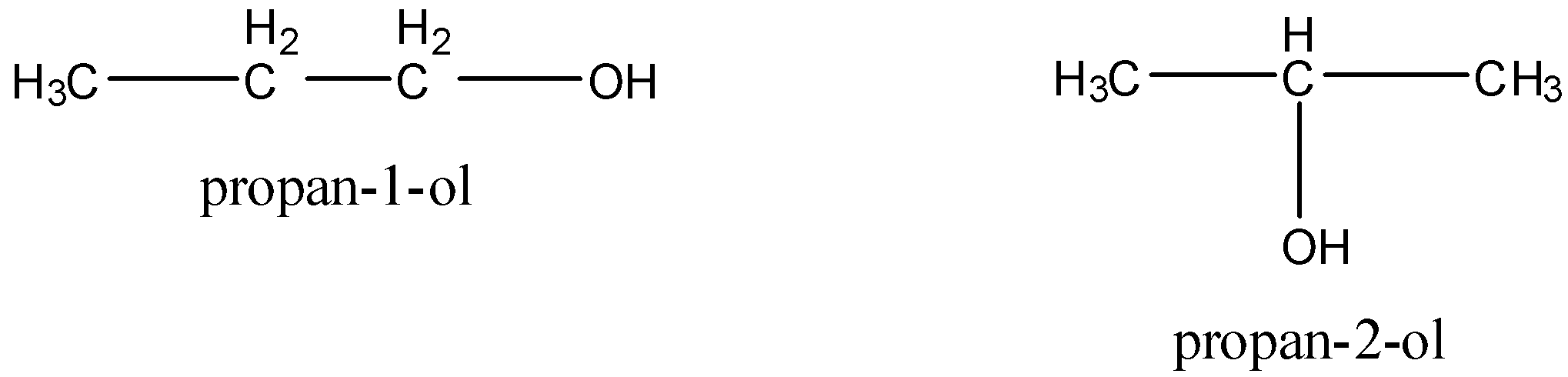
Now, we draw the structure of dimethyl ether and ethyl alcohol.

So, both the compounds differ by their functional groups. So, they are functional isomers.
Therefore, the correct answer is Option C.
Note: Tautomers are actually functional isomers which exist simultaneously and also in dynamic equilibrium. The isomerism in this case is termed as tautomerism. It is of different types but the most common among them is the keto-enol tautomerism. This isomerism arises due to 1,3 migration of the hydrogen atom from carbon to oxygen atom and vice versa. For example,

Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




