Cyanohydrin of which compound on hydrolysis will give lactic acid?
A. \[{{\rm{C}}_6}{{\rm{H}}_5}{\rm{CHO}}\]
B. \[{\rm{HCHO}}\]
C. \[{\rm{C}}{{\rm{H}}_3}{\rm{CHO}}\]
D. \[{\rm{C}}{{\rm{H}}_3}{\rm{C}}{{\rm{H}}_2}{\rm{CHO}}\]
Answer
602.4k+ views
Hint: Lactic acid contains three carbon atoms. It is obtained from the cyanohydrin of an aldehyde. The number of carbon atoms in the cyanohydrin will be the same as the number of carbon atoms in lactic acid. But the starting aldehyde will have one less carbon atom, as one carbon comes from the cyano group.
Complete step by step answer:
The aldehydes react with hydrogen cyanide to form cyanohydrin.

In the above reaction, ‘R’ represents an alkyl group. The cyanohydrin has an additional carbon atom than aldehyde. This additional carbon atom comes from hydrogen cyanide. The cyano group of hydrogen cyanide is added to carbonyl carbon and the hydrogen atom of hydrogen cyanide is added to carbonyl oxygen. A new carbon-carbon single bond is formed and the carbon-oxygen double bond is converted into a single bond. Aldehyde contains carbonyl groups whereas cyanohydrin contains one hydroxyl group and one cyano group.
The hydrolysis of cyanohydrin gives alpha hydroxy, carboxylic acids.
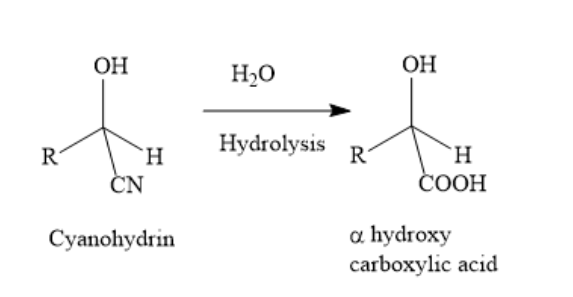
In the above reaction, the cyano group is hydrolyzed to a carboxylic acid functional group. The number of carbon atoms in the alpha hydroxy carboxylic acid is the same as the number of carbon atoms in the cyanohydrin.
When an alpha hydroxy carboxylic acid is given and it is asked to determine the structure of aldehyde, whose cyanohydrin upon hydrolysis will give the given alpha hydroxy carboxylic acid, then, decrease the number of carbon atoms of the alpha hydroxy carboxylic acid by one to obtain the number of carbon atoms of the aldehyde.
structure of lactic acid is as shown below:
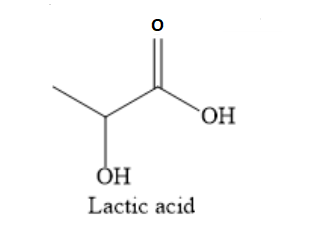
Lactic acid contains 3 carbon atoms. The corresponding aldehyde will have 3-1=2 carbon atoms.
In the given options for aldehydes, the number of carbon atoms in benzaldehyde, formaldehyde, acetaldehyde and propionaldehyde is 7,1, 2, and 3 respectively. Acetaldehyde contains 2 carbon atoms.
Hence, option C) \[{\rm{C}}{{\rm{H}}_3}{\rm{CHO}}\] is the correct answer.
Note:
Since cyano group is added to carbonyl group, cyanohydrin has one excess carbon atom than corresponding aldehyde. Hence, while determining the structure of the starting aldehyde, do not forget to account for additional carbon atoms from the cyano group.
Complete step by step answer:
The aldehydes react with hydrogen cyanide to form cyanohydrin.

In the above reaction, ‘R’ represents an alkyl group. The cyanohydrin has an additional carbon atom than aldehyde. This additional carbon atom comes from hydrogen cyanide. The cyano group of hydrogen cyanide is added to carbonyl carbon and the hydrogen atom of hydrogen cyanide is added to carbonyl oxygen. A new carbon-carbon single bond is formed and the carbon-oxygen double bond is converted into a single bond. Aldehyde contains carbonyl groups whereas cyanohydrin contains one hydroxyl group and one cyano group.
The hydrolysis of cyanohydrin gives alpha hydroxy, carboxylic acids.

In the above reaction, the cyano group is hydrolyzed to a carboxylic acid functional group. The number of carbon atoms in the alpha hydroxy carboxylic acid is the same as the number of carbon atoms in the cyanohydrin.
When an alpha hydroxy carboxylic acid is given and it is asked to determine the structure of aldehyde, whose cyanohydrin upon hydrolysis will give the given alpha hydroxy carboxylic acid, then, decrease the number of carbon atoms of the alpha hydroxy carboxylic acid by one to obtain the number of carbon atoms of the aldehyde.
structure of lactic acid is as shown below:

Lactic acid contains 3 carbon atoms. The corresponding aldehyde will have 3-1=2 carbon atoms.
In the given options for aldehydes, the number of carbon atoms in benzaldehyde, formaldehyde, acetaldehyde and propionaldehyde is 7,1, 2, and 3 respectively. Acetaldehyde contains 2 carbon atoms.
Hence, option C) \[{\rm{C}}{{\rm{H}}_3}{\rm{CHO}}\] is the correct answer.
Note:
Since cyano group is added to carbonyl group, cyanohydrin has one excess carbon atom than corresponding aldehyde. Hence, while determining the structure of the starting aldehyde, do not forget to account for additional carbon atoms from the cyano group.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




