What is the correct name of $I{{F}_{7}}$.
(A) Iron heptafluoride.
(B) Iodine heptafluoride
(C) Iodine septafluroide
(D) Iron (IV) septafluoride.
Answer
613.8k+ views
Hint: $I{{F}_{7}}$ is the best example for interhalogen compounds. Interhalogen compounds mean halogens themselves react to each other and form interhalogen compounds. $I{{F}_{7}}$ has a structure of pentagonal bipyramidal.
Complete step by step solution:
-In $I{{F}_{7}}$, one iodine and seven fluorine atoms are present.
-The chemical name of $I{{F}_{7}}$ is Iodine heptafluoride.
-In the chemical name of $I{{F}_{7}}$ the name of Iodine should be written first because of its less electronegativity. Fluorine's name should be written later because it is more electronegativity.
-The prefix hepta in the name of $I{{F}_{7}}$ - Iodine heptafluoride indicates the presence of seven fluorine atoms.
So, the correct option is (B).
Additional information:
-The structure of $I{{F}_{7}}$ is as follows.
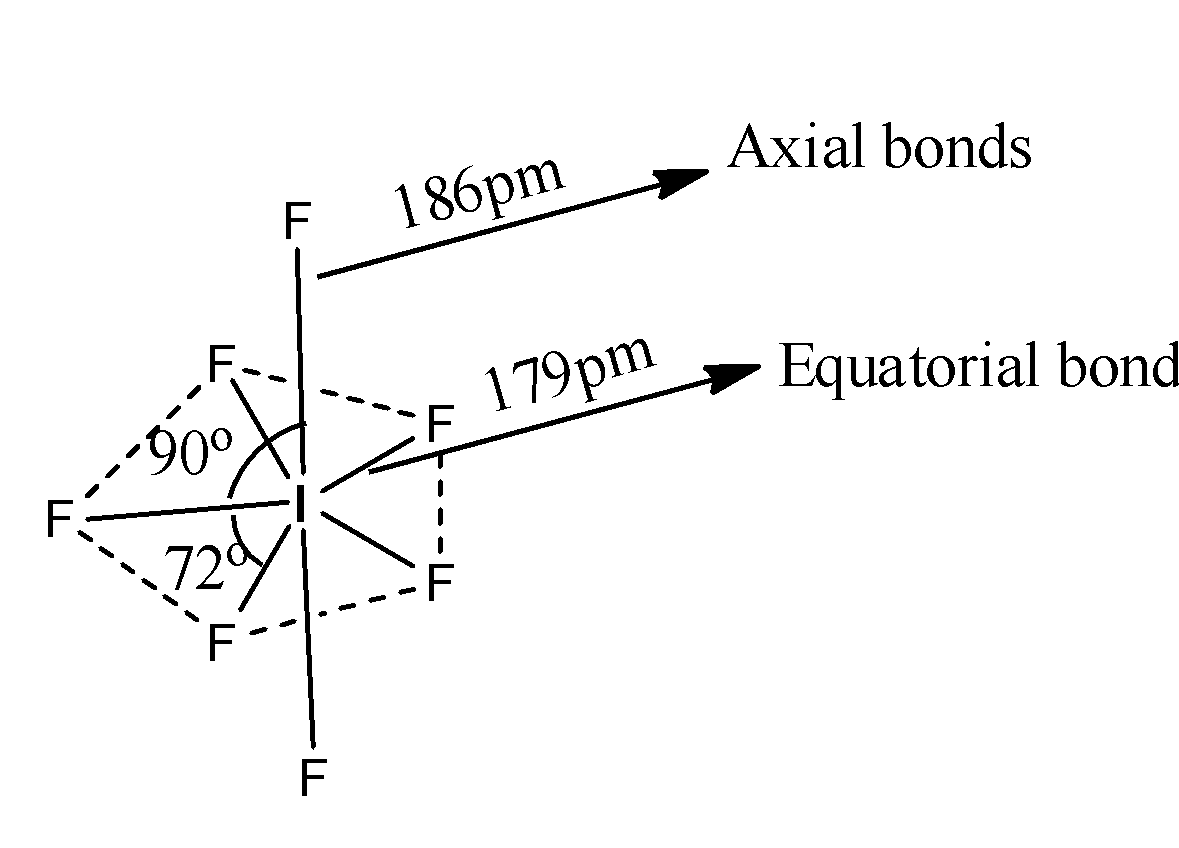
-The resultant geometry of $I{{F}_{7}}$ is pentagonal bipyramidal.
-The hybridization of Iodine in $I{{F}_{7}}$ is $s{{p}^{3}}{{d}^{3}}$ and the overlapping of fluorine with iodine orbitals we can see as follows.
-There are two types of bonds axial and equatorial bonds.
-Axial bonds are longer than equatorial bonds.
-There are two types of orthogonal angles, there are 90 and 72.
Note: If any chemical contains a number of repeating atoms in its chemical formula then we are not supposed to write the number in the chemical name. If the compound contains Two same atoms – di, three same atoms – tri, four same atoms – tetra, five same atoms – penta, six same atoms – hexa, seven same atoms – septa, eight same atoms – octa.
Complete step by step solution:
-In $I{{F}_{7}}$, one iodine and seven fluorine atoms are present.
-The chemical name of $I{{F}_{7}}$ is Iodine heptafluoride.
-In the chemical name of $I{{F}_{7}}$ the name of Iodine should be written first because of its less electronegativity. Fluorine's name should be written later because it is more electronegativity.
-The prefix hepta in the name of $I{{F}_{7}}$ - Iodine heptafluoride indicates the presence of seven fluorine atoms.
So, the correct option is (B).
Additional information:
-The structure of $I{{F}_{7}}$ is as follows.

-The resultant geometry of $I{{F}_{7}}$ is pentagonal bipyramidal.
-The hybridization of Iodine in $I{{F}_{7}}$ is $s{{p}^{3}}{{d}^{3}}$ and the overlapping of fluorine with iodine orbitals we can see as follows.
-There are two types of bonds axial and equatorial bonds.
-Axial bonds are longer than equatorial bonds.
-There are two types of orthogonal angles, there are 90 and 72.
Note: If any chemical contains a number of repeating atoms in its chemical formula then we are not supposed to write the number in the chemical name. If the compound contains Two same atoms – di, three same atoms – tri, four same atoms – tetra, five same atoms – penta, six same atoms – hexa, seven same atoms – septa, eight same atoms – octa.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




