What is the correct IUPAC name of this compound?
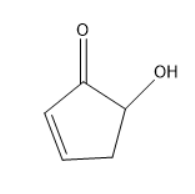
I’m not able to understand why the name of this compound is \[5 - hydroxycyclopent - 2 - enone\] and not \[2 - hydroxycyclopent - 4 - enone\] . The \[ - OH\] groups have a higher priority than the alkene group so it should be given a smaller number than alkene and hence the name should be \[2 - hydroxycyclopent - 4 - enone\] .

Answer
525.6k+ views
Hint: Chemical compounds are classified into functional groups based on the groups. Ketones are the carbonyl compounds that have \[C = O\] group which has higher priority than alcohols, the compounds containing \[ - OH\] groups. When double bond is nearer to ketones, then the numbering should be done on the side of double bond only.
Complete answer:
There were so many chemical compounds in chemistry whose structures were very similar. IUPAC nomenclature distinguishes very similar structures by the position of groups. IUPAC stands for international union of pure and applied chemistry.
When the molecule consists of a double bond, ketone functional group, and alcohol functional group. Then the priority should be given to ketones rather than alcohols. Thus, the numbering should start from the ketone group only. As the double bond is nearer to the ketone, the numbering should go in the direction of the double bond only according to IUPAC nomenclature.
Thus, the IUPAC name of the compound is \[5 - hydroxycyclopent - 2 - enone\] but not \[2 - hydroxycyclopent - 4 - enone\] .
Note:
There were so many rules while writing the IUPAC nomenclature of the chemical compounds. The ketone has higher priority than alcohols is an important point, and the double bond must get lower number when the double bond is nearer to the ketone functional group though alcoholic functional group is present.
Complete answer:
There were so many chemical compounds in chemistry whose structures were very similar. IUPAC nomenclature distinguishes very similar structures by the position of groups. IUPAC stands for international union of pure and applied chemistry.
When the molecule consists of a double bond, ketone functional group, and alcohol functional group. Then the priority should be given to ketones rather than alcohols. Thus, the numbering should start from the ketone group only. As the double bond is nearer to the ketone, the numbering should go in the direction of the double bond only according to IUPAC nomenclature.
Thus, the IUPAC name of the compound is \[5 - hydroxycyclopent - 2 - enone\] but not \[2 - hydroxycyclopent - 4 - enone\] .
Note:
There were so many rules while writing the IUPAC nomenclature of the chemical compounds. The ketone has higher priority than alcohols is an important point, and the double bond must get lower number when the double bond is nearer to the ketone functional group though alcoholic functional group is present.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




