How to convert Aniline to nitrobenzene?
Answer
578.1k+ views
Hint: Aniline cannot be directly converted to nitrobenzene directly. It needs to be converted to a diazonium salt first which can then be treated with other chemical agents to form the nitrobenzene.
Complete Stepwise Solution:
Aniline is a basic molecule which comprises an amino group attached to the benzene ring. This can be treated with a cold dilute acidic solution sodium nitrite in the temperature range of $ {\left( {{\text{0 - 5}}} \right)^{{\text{ 0}}}}{\text{C}} $ to form diazo compounds which form the intermediate for the transformation reaction. This is then treated with hydrogen fluoroborate or $ {\text{HB}}{{\text{F}}_{\text{4}}} $ to form the fluro salt of the diazo compound. Then this salt is treated with copper and sodium nitrite to form nitrobenzene. The reaction can be written as follows:
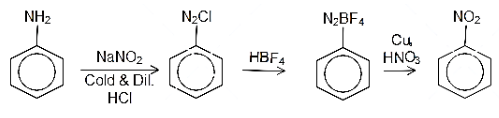
Alternatively, there is one more route for the preparation of nitrobenzene from aniline. aniline can be treated with a cold dilute acidic solution sodium nitrite in the temperature range of $ {\left( {{\text{0 - 5}}} \right)^{{\text{ 0}}}}{\text{C}} $ to form diazo compound. Then diazonium chloride is then hydrolysed in presence of hypophosphorous acid and water to form benzene. The benzene is then treated with concentrated solution of sulphuric acid and nitric acid to form the nitrobenzene. The reaction can be written as follows:
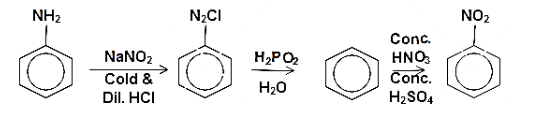
Note:
The reaction of Aniline with the cold dilute acidic solution of sodium nitrate to form the diazonium salt serves to be an intermediate for the preparation of a number of compounds. The diazo salt is not stable at high temperatures and hence the reaction takes place at cold temperature conditions.
Complete Stepwise Solution:
Aniline is a basic molecule which comprises an amino group attached to the benzene ring. This can be treated with a cold dilute acidic solution sodium nitrite in the temperature range of $ {\left( {{\text{0 - 5}}} \right)^{{\text{ 0}}}}{\text{C}} $ to form diazo compounds which form the intermediate for the transformation reaction. This is then treated with hydrogen fluoroborate or $ {\text{HB}}{{\text{F}}_{\text{4}}} $ to form the fluro salt of the diazo compound. Then this salt is treated with copper and sodium nitrite to form nitrobenzene. The reaction can be written as follows:

Alternatively, there is one more route for the preparation of nitrobenzene from aniline. aniline can be treated with a cold dilute acidic solution sodium nitrite in the temperature range of $ {\left( {{\text{0 - 5}}} \right)^{{\text{ 0}}}}{\text{C}} $ to form diazo compound. Then diazonium chloride is then hydrolysed in presence of hypophosphorous acid and water to form benzene. The benzene is then treated with concentrated solution of sulphuric acid and nitric acid to form the nitrobenzene. The reaction can be written as follows:

Note:
The reaction of Aniline with the cold dilute acidic solution of sodium nitrate to form the diazonium salt serves to be an intermediate for the preparation of a number of compounds. The diazo salt is not stable at high temperatures and hence the reaction takes place at cold temperature conditions.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




