How can I compare the structural formula and sawhorse projection of 2-bromo-3-chlorobutane?
Answer
565.5k+ views
Hint: Stereoisomers of compounds have the ability to have a spatial arrangement in space and are called conformers. They have a free rotation of carbon that arranges in different orientations in space giving us various projections.
Complete answer:
The compound 2-bromo-3-chlorobutane contains a 4-carbon chain along with bromide and chloride group at adjacent, 2 and 3 carbons respectively.
The structural formula of 2-bromo-3-chlorobutane can be made as:
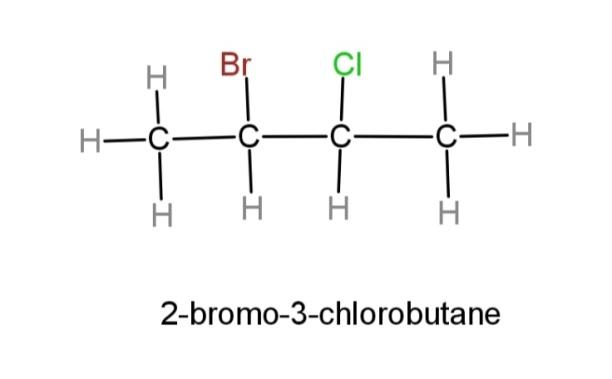
The structural formula is a straight chain of carbon along with bromide and chloride groups. This structure does not tell us about any stereochemical aspect of the compound, which is its spatial arrangement in space.
Sawhorse projection is a method to show the spatial arrangement of atoms in a compound as follows:
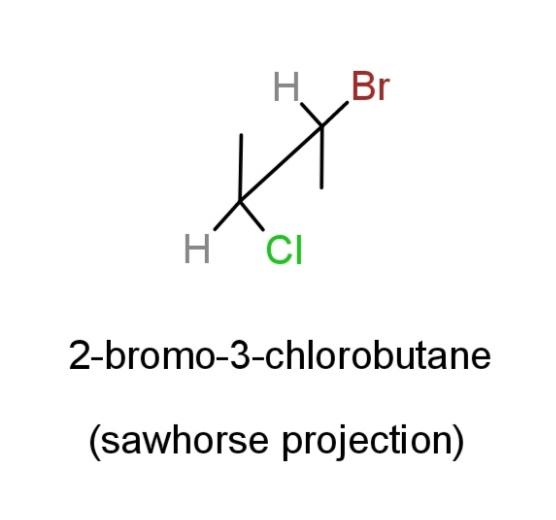
This sawhorse projection structure tells us that the methyl groups are attached at both the ends, showing the bond at the vertical line. While the bromide group is attached at carbon-2, and chloride group at carbon-3. This also shows that carbon-2 and carbon-3 have chirality and therefore, four stereoisomers can be derived from 2-bromo-3-chlorobutane.
Hence, the structural formula of 2-bromo-3-chlorobutane does not tell any of its stereochemistry, while the sawhorse projection structure tells us the stereochemistry of this compound.
Note:
Sawhorse projection can be of two types, staggered projection and eclipsed projection. Here eclipsed projection is shown. The projection becomes staggered when both the end group lines are vertically upwards.
Complete answer:
The compound 2-bromo-3-chlorobutane contains a 4-carbon chain along with bromide and chloride group at adjacent, 2 and 3 carbons respectively.
The structural formula of 2-bromo-3-chlorobutane can be made as:

The structural formula is a straight chain of carbon along with bromide and chloride groups. This structure does not tell us about any stereochemical aspect of the compound, which is its spatial arrangement in space.
Sawhorse projection is a method to show the spatial arrangement of atoms in a compound as follows:

This sawhorse projection structure tells us that the methyl groups are attached at both the ends, showing the bond at the vertical line. While the bromide group is attached at carbon-2, and chloride group at carbon-3. This also shows that carbon-2 and carbon-3 have chirality and therefore, four stereoisomers can be derived from 2-bromo-3-chlorobutane.
Hence, the structural formula of 2-bromo-3-chlorobutane does not tell any of its stereochemistry, while the sawhorse projection structure tells us the stereochemistry of this compound.
Note:
Sawhorse projection can be of two types, staggered projection and eclipsed projection. Here eclipsed projection is shown. The projection becomes staggered when both the end group lines are vertically upwards.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




