\[{[{\text{Co}}{{\text{F}}_6}]^{3 - }}\]
is a coordination complex ion. How many unpaired electrons are there in the complex?
Answer
630.6k+ views
Hint: F is a weak field ligand which causes small crystal field splitting. Weak ligands lead to formation of high spin complexes.
Complete answer:
We will first calculate the oxidation state of \[{\text{Co}}\]
in \[{[{\text{Co}}{{\text{F}}_6}]^{3 - }}\]
We take the oxidation state of \[{\text{Co}}\]
as ‘x’ and the charge on one is \[ - 1\]. Total charge on the complex is \[{\text{ - 3}}\].
So, \[{\text{x + ( - 6) = - 3}}\]
\[{\text{ x = - 3 + 6}}\]
\[{\text{x = + 3}}\]
The outer electronic configuration of \[{\text{Co}}\]is \[[{\text{Ar] 3}}{{\text{d}}^7}{\text{4}}{{\text{s}}^2}\].
So, in \[{\text{C}}{{\text{o}}^{ + 3}}\]it becomes \[[{\text{Ar] 3}}{{\text{d}}^6}\].
Since F is a weak field ligand, it will lead to splitting of d orbitals. First it leads to \[{{\text{t}}_{2g}}\]to be half filled and then \[{e_g}\]to be half filled too and then pairing will occur.
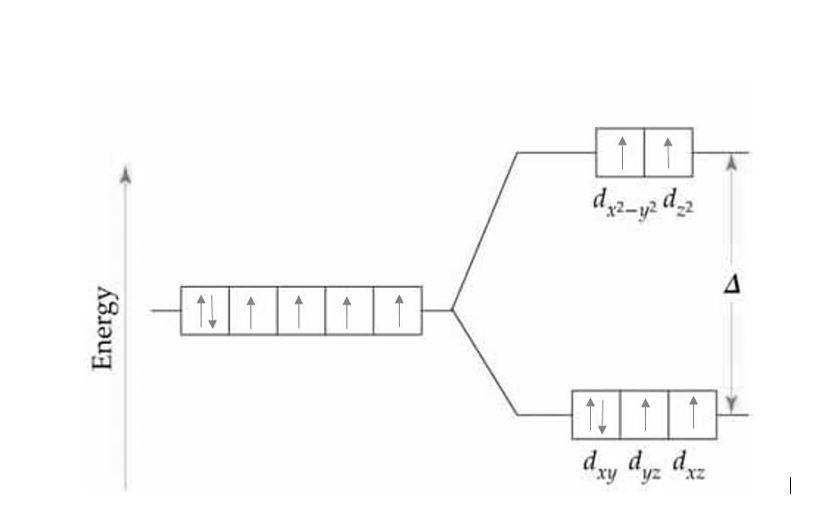
Hence, there will be 4 unpaired electrons.
Additional information: Crystal field splitting describes breaking of orbitals in complex formed by transition metal ions due to ligands. It considers the bond between metal and ligand to be purely ionic. Theory says that this bond arises due to electrostatic interactions between metal and the ligand. In case of anionic complexes, ligands are considered as point charges. It is a qualitative measure of the strength of metal-ligand bond.
\[{[{\text{Co}}{{\text{F}}_6}]^{3 - }}\]is a high spin complex since fluorine is a weak field ligand that causes small splitting. On the other hand, ligands that cause large crystal field splitting, result in formation of low spin complexes and are called strong ligands.
Note: To remember this easily, we will keep in mind that weak field ligands form high spin complexes and strong field ligands form low spin complexes.
Complete answer:
We will first calculate the oxidation state of \[{\text{Co}}\]
in \[{[{\text{Co}}{{\text{F}}_6}]^{3 - }}\]
We take the oxidation state of \[{\text{Co}}\]
as ‘x’ and the charge on one is \[ - 1\]. Total charge on the complex is \[{\text{ - 3}}\].
So, \[{\text{x + ( - 6) = - 3}}\]
\[{\text{ x = - 3 + 6}}\]
\[{\text{x = + 3}}\]
The outer electronic configuration of \[{\text{Co}}\]is \[[{\text{Ar] 3}}{{\text{d}}^7}{\text{4}}{{\text{s}}^2}\].
So, in \[{\text{C}}{{\text{o}}^{ + 3}}\]it becomes \[[{\text{Ar] 3}}{{\text{d}}^6}\].
Since F is a weak field ligand, it will lead to splitting of d orbitals. First it leads to \[{{\text{t}}_{2g}}\]to be half filled and then \[{e_g}\]to be half filled too and then pairing will occur.

Hence, there will be 4 unpaired electrons.
Additional information: Crystal field splitting describes breaking of orbitals in complex formed by transition metal ions due to ligands. It considers the bond between metal and ligand to be purely ionic. Theory says that this bond arises due to electrostatic interactions between metal and the ligand. In case of anionic complexes, ligands are considered as point charges. It is a qualitative measure of the strength of metal-ligand bond.
\[{[{\text{Co}}{{\text{F}}_6}]^{3 - }}\]is a high spin complex since fluorine is a weak field ligand that causes small splitting. On the other hand, ligands that cause large crystal field splitting, result in formation of low spin complexes and are called strong ligands.
Note: To remember this easily, we will keep in mind that weak field ligands form high spin complexes and strong field ligands form low spin complexes.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




