Choose the correct sequence for the geometry of the given molecule: borazone, borazole, ${B_3}{O_6}^{ - 3}$ ,$Fe_2Cl_6$ and trimer of $FCN$.
[P stands for planar, NP stands for Non planar]
A.NP,NP,P,P,P
B.P,P,NP,NP,P
C.NP,NP,P,NP,P
D.NP,P,P,NP,P
Answer
588.3k+ views
Hint: it is required to find out the electronic configuration of the central atom of these compounds. After finding the electronic configuration it is required to decide the hybridization of the compound. Mostly all \[{s}{{{p}}^{{3}}},\text{ }{s}{{{p}}^{{3}}}{d},\text{ }{s}{{{p}}^{{3}}}{{{d}}^{{2}}},\text{ }{s}{{{p}}^{{3}}}{{{d}}^{{3}}}\] hybridized compounds are non-planar.
Complete step by step answer:
Taking each molecule one by one we shall examine whether they are planar or not.
Borazone in general is cubic boron nitride. it has the same structure as that of a diamond. If we can recall the hybridization of diamond is $s{{p}^{3}}$this means that just like the diamond, the geometry of borazone is also non planar.
Borazole is also known as inorganic benzene. It has a structure similar to benzene except in place of carbon atoms we find boron atoms thus earning the name inorganic benzene. The hybridization of carbon in benzene is $s{{p}^{2}}$ and therefore, comparing the structures we can say that the hybridization of borazole being the same will mean that it is indeed, planar. The molecular formula is $B_3N_3H_6$.
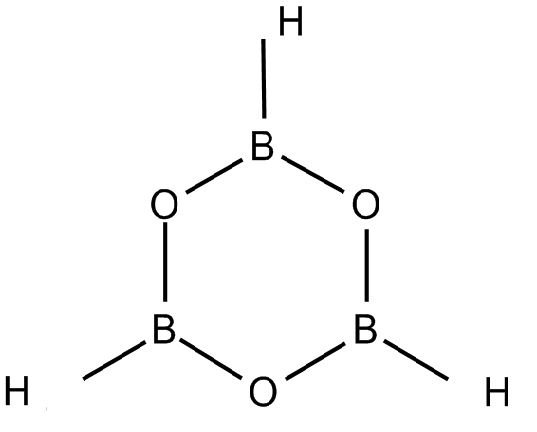
${B_3}{O_6}^{ - 3}$ is combined with hydrogen ions to form $H_3B_3O_6$. This compound is known as boroxine. It is a planar molecule as the boron in this compound is joined to two oxygen atoms as shown in the diagram below.
$Fe_2Cl_6$is a dimer of ferric chloride. it is non planar. The dimer is connected to each other through a bond known as chloride bridges which means that some bonds may be out of the main plane.
Trimer of $FCN$ which is basically three molecules of carbononitridic fluoride also known as cyanogen fluoride. It forms a ring structure during trimerization. It is similar to the structure of borazole thus making it a planar molecule.
So, the correct answer is Option D.
Note: the important point to remember is that a particular compound is said to be non planar when the surrounding atoms bonded to the central atom are not in the same plane. the surrounding atoms are usually perpendicular to the central atom. Also remember that mostly all \[{s}{{{p}}^{{3}}},\text{ }{s}{{{p}}^{{3}}}{d},\text{ }{s}{{{p}}^{{3}}}{{{d}}^{{2}}},\text{ }{s}{{{p}}^{{3}}}{{{d}}^{{3}}}\]hybridized compounds are non-planar.
Complete step by step answer:
Taking each molecule one by one we shall examine whether they are planar or not.
Borazone in general is cubic boron nitride. it has the same structure as that of a diamond. If we can recall the hybridization of diamond is $s{{p}^{3}}$this means that just like the diamond, the geometry of borazone is also non planar.
Borazole is also known as inorganic benzene. It has a structure similar to benzene except in place of carbon atoms we find boron atoms thus earning the name inorganic benzene. The hybridization of carbon in benzene is $s{{p}^{2}}$ and therefore, comparing the structures we can say that the hybridization of borazole being the same will mean that it is indeed, planar. The molecular formula is $B_3N_3H_6$.
${B_3}{O_6}^{ - 3}$ is combined with hydrogen ions to form $H_3B_3O_6$. This compound is known as boroxine. It is a planar molecule as the boron in this compound is joined to two oxygen atoms as shown in the diagram below.

$Fe_2Cl_6$is a dimer of ferric chloride. it is non planar. The dimer is connected to each other through a bond known as chloride bridges which means that some bonds may be out of the main plane.
Trimer of $FCN$ which is basically three molecules of carbononitridic fluoride also known as cyanogen fluoride. It forms a ring structure during trimerization. It is similar to the structure of borazole thus making it a planar molecule.
So, the correct answer is Option D.
Note: the important point to remember is that a particular compound is said to be non planar when the surrounding atoms bonded to the central atom are not in the same plane. the surrounding atoms are usually perpendicular to the central atom. Also remember that mostly all \[{s}{{{p}}^{{3}}},\text{ }{s}{{{p}}^{{3}}}{d},\text{ }{s}{{{p}}^{{3}}}{{{d}}^{{2}}},\text{ }{s}{{{p}}^{{3}}}{{{d}}^{{3}}}\]hybridized compounds are non-planar.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE




