How many bonding orbitals are in silicon tetrafluoride or $\text{Si}{{\text{F}}_{\text{4}}}$?
Answer
579k+ views
Hint: In silicon tetrafluoride, there are four bonding orbital. Bounding orbital as a name indicates the attractive interaction between the atomic orbital of two or more atoms in molecules, electrons in the orbitals Stabilize a molecule.
Complete step by step answer:
Silicon tetrafluoride is a covalent molecule. It contains a central silicon atom which is bound to four fluorine atoms through four single covalent bonds. Silicon has four unpaired electrons, To fulfill its valence silicon atom gets attached to four fluorine atoms. Fluorine atoms contain one valence electron to form a covalent bond with silicon.
$4$ bonding pairs with zero non-bonding pairs on silicon atom results in a tetrahedral shape of ${{\text{S}}_{\text{1}}}{{\text{F}}_{\text{4}}}$.
Lewis Structure of $\text{Si}{{\text{F}}_{4}}$
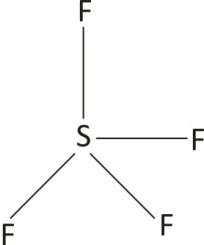
Since each single covalent bond represent a $\sigma $ orbitals there are four bonding orbitals in $\text{Si}{{\text{F}}_{4}}$
$\text{Si}{{\text{F}}_{4}}$ have $s{{p}^{3}}$ hybridization.
The bonding orbital is defined as an attractive interaction between the atomic orbital of a bonding atom in a molecule.
Note: Silicon tetrafluoride is a colorless gas with strong odor.
It is used to seal oil well from water and also used in production of silicon and fluosilicic acid.
Silicon tetrafluoride is hazardous.
Complete step by step answer:
Silicon tetrafluoride is a covalent molecule. It contains a central silicon atom which is bound to four fluorine atoms through four single covalent bonds. Silicon has four unpaired electrons, To fulfill its valence silicon atom gets attached to four fluorine atoms. Fluorine atoms contain one valence electron to form a covalent bond with silicon.
$4$ bonding pairs with zero non-bonding pairs on silicon atom results in a tetrahedral shape of ${{\text{S}}_{\text{1}}}{{\text{F}}_{\text{4}}}$.
Lewis Structure of $\text{Si}{{\text{F}}_{4}}$

Since each single covalent bond represent a $\sigma $ orbitals there are four bonding orbitals in $\text{Si}{{\text{F}}_{4}}$
$\text{Si}{{\text{F}}_{4}}$ have $s{{p}^{3}}$ hybridization.
The bonding orbital is defined as an attractive interaction between the atomic orbital of a bonding atom in a molecule.
Note: Silicon tetrafluoride is a colorless gas with strong odor.
It is used to seal oil well from water and also used in production of silicon and fluosilicic acid.
Silicon tetrafluoride is hazardous.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




