What is the Bohr-Bury scheme of arrangement of electrons in an atom?
Answer
520.5k+ views
Hint: Electronic configuration of an atom is governed by a number of rules which includes: Aufbau principle, Bohr-Bury rule, Pauli exclusion principle and Hund’s rule. All these rules are employed to write the correct electronic configuration of any atom.
Complete answer:
Bohr-Bury rules are used to explain some important aspects of writing electronic configuration. According to this rule-
The energy of any orbital is determined with the help of numbers $n$ and $l$.
The filling of electrons in the orbital takes place according to the increasing order of $\left( {n + l} \right)$.
For example: energy of $3d$ orbital is $\left( {3 + 2 = 5} \right)$, while the energy of $4s$ orbital is $\left( {4 + 0 = 4} \right)$. Hence, according to Bohr-Bury rules energy of $3d$ is more than $4s$ orbital hence, electrons first fill in $3d$ orbital then fill into $4s$ orbital.
In case, if two orbitals possess the same energy level then the electron fills into an orbital which is associated with a lower value of $n$.
For example: the energy level of $2p$ orbital is $\left( {2 + 1 = 3} \right)$, and the energy level of $3s$ orbital is $\left( {3 + 1 = 3} \right)$. In this case the energy level of both the orbital is the same but electron first fills into $2p$ orbital because the value of $n$ is smaller in $2p$.
This rule helps in identifying the fact that an orbital with higher value of principal quantum number $n$, may have lower energy level than orbitals with lower value of principal quantum number.
Bohr-Bury states that the maximum capacity of a shell to hold electrons in them is equal to $2{n^2}$. Where $n$ describes the orbital energy of electrons.
Outermost shell of an atom must contain a maximum of $8$ electrons to attain stability.
Electron filling proceeds to another shell until its inner shell is completely filled.
Bohr-Bury scheme of arrangement of electrons in an atom is shown as:
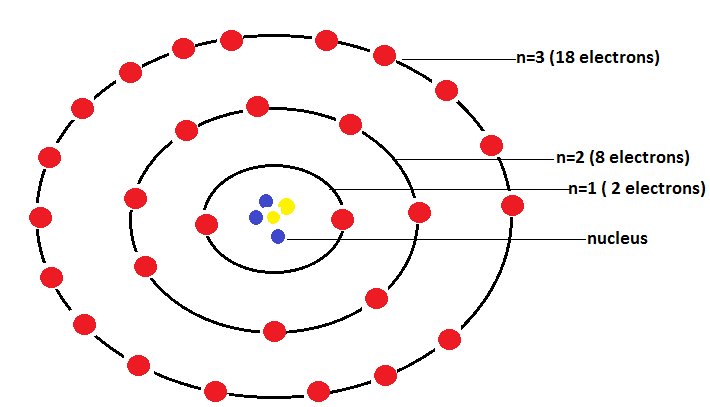
Note:
Aufbau helps in determining the energy of orbitals while Pauli explains the spin and maximum holding capacity of an orbital. Remember that a maximum of $2$ electrons must be present in one orbital while a maximum of $8$ present in orbit.
Complete answer:
Bohr-Bury rules are used to explain some important aspects of writing electronic configuration. According to this rule-
The energy of any orbital is determined with the help of numbers $n$ and $l$.
The filling of electrons in the orbital takes place according to the increasing order of $\left( {n + l} \right)$.
For example: energy of $3d$ orbital is $\left( {3 + 2 = 5} \right)$, while the energy of $4s$ orbital is $\left( {4 + 0 = 4} \right)$. Hence, according to Bohr-Bury rules energy of $3d$ is more than $4s$ orbital hence, electrons first fill in $3d$ orbital then fill into $4s$ orbital.
In case, if two orbitals possess the same energy level then the electron fills into an orbital which is associated with a lower value of $n$.
For example: the energy level of $2p$ orbital is $\left( {2 + 1 = 3} \right)$, and the energy level of $3s$ orbital is $\left( {3 + 1 = 3} \right)$. In this case the energy level of both the orbital is the same but electron first fills into $2p$ orbital because the value of $n$ is smaller in $2p$.
This rule helps in identifying the fact that an orbital with higher value of principal quantum number $n$, may have lower energy level than orbitals with lower value of principal quantum number.
Bohr-Bury states that the maximum capacity of a shell to hold electrons in them is equal to $2{n^2}$. Where $n$ describes the orbital energy of electrons.
Outermost shell of an atom must contain a maximum of $8$ electrons to attain stability.
Electron filling proceeds to another shell until its inner shell is completely filled.
Bohr-Bury scheme of arrangement of electrons in an atom is shown as:

Note:
Aufbau helps in determining the energy of orbitals while Pauli explains the spin and maximum holding capacity of an orbital. Remember that a maximum of $2$ electrons must be present in one orbital while a maximum of $8$ present in orbit.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




