Why the blue color of copper sulfate solution in water changes to yellow when treated with concentrated hydrochloric acid?
Answer
631.5k+ views
Hint: Color can be changed because of the change in ligands. It is due to crystal field splitting. The number of unpaired electrons in the last subshell is counted.
Complete step by step answer:
Copper is a d-block element and most of the transition metal compounds are colored both in the solid-state and in aqueous solution.
Color is due to the presence of incomplete d-subshell. Further, when the anion or the ligands approach the transition metal ions, their d-orbitals do not remain degenerated. They split into two sets, one consisting of lower energy orbitals (\[{{\text{t}}_{\text{2g}}}\text{which includes }{{\text{d}}_{\text{xy}}}\text{,}{{\text{d}}_{\text{yz}}}\text{, and }{{\text{d}}_{\text{zx}}}\]) and the other consisting of higher energy orbitals (\[{{e}_{\text{g}}}\text{which includes }{{\text{d}}_{{{\text{x}}^{2}}-{{y}^{2}}}}\text{, and }{{\text{d}}_{{{\text{z}}^{2}}}}\]). This is called crystal field splitting.
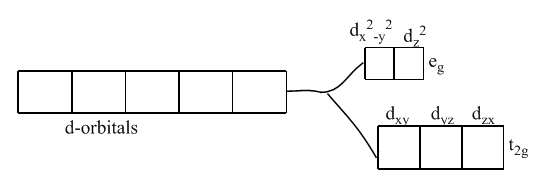
Thus, the electron can jump from lower energy d-orbital to higher energy d-orbital. The required amount of energy to do this is obtained by absorption of light of a particular wavelength in the region of visible light. The transition metal ion has the property to absorb such radiations from the visible light and appears colored due to the emission of the remainder as colored light.
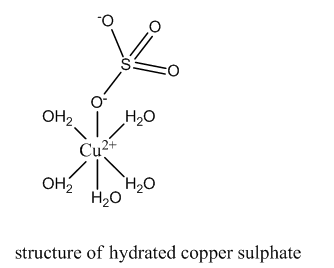
In hydrated copper sulfate, four water molecules are present as ligands. In the presence of these ligands, d-orbital are no longer degenerate in energy. Hence, the d-d transition takes place absorbing red wavelength. The complementary color, viz., blue is reflected.
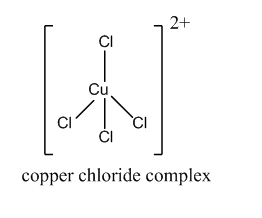
But when concentrated hydrochloric acid is added to the hydrated copper sulfate, the water ligands are replaced by chloride ligands. This forms the copper chloride complex. The d-d transition takes place by absorbing the violet wavelength. Hence, the reflection is yellow.
Hint: There are specific colors of specific ligands that they will impart. Don’t get confused between the wavelength absorbed and wavelength emitted. The color of the compound will always be complementary to the color of the wavelength absorbed.
Complete step by step answer:
Copper is a d-block element and most of the transition metal compounds are colored both in the solid-state and in aqueous solution.
Color is due to the presence of incomplete d-subshell. Further, when the anion or the ligands approach the transition metal ions, their d-orbitals do not remain degenerated. They split into two sets, one consisting of lower energy orbitals (\[{{\text{t}}_{\text{2g}}}\text{which includes }{{\text{d}}_{\text{xy}}}\text{,}{{\text{d}}_{\text{yz}}}\text{, and }{{\text{d}}_{\text{zx}}}\]) and the other consisting of higher energy orbitals (\[{{e}_{\text{g}}}\text{which includes }{{\text{d}}_{{{\text{x}}^{2}}-{{y}^{2}}}}\text{, and }{{\text{d}}_{{{\text{z}}^{2}}}}\]). This is called crystal field splitting.

Thus, the electron can jump from lower energy d-orbital to higher energy d-orbital. The required amount of energy to do this is obtained by absorption of light of a particular wavelength in the region of visible light. The transition metal ion has the property to absorb such radiations from the visible light and appears colored due to the emission of the remainder as colored light.
In hydrated copper sulfate, four water molecules are present as ligands. In the presence of these ligands, d-orbital are no longer degenerate in energy. Hence, the d-d transition takes place absorbing red wavelength. The complementary color, viz., blue is reflected.

But when concentrated hydrochloric acid is added to the hydrated copper sulfate, the water ligands are replaced by chloride ligands. This forms the copper chloride complex. The d-d transition takes place by absorbing the violet wavelength. Hence, the reflection is yellow.

Hint: There are specific colors of specific ligands that they will impart. Don’t get confused between the wavelength absorbed and wavelength emitted. The color of the compound will always be complementary to the color of the wavelength absorbed.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




