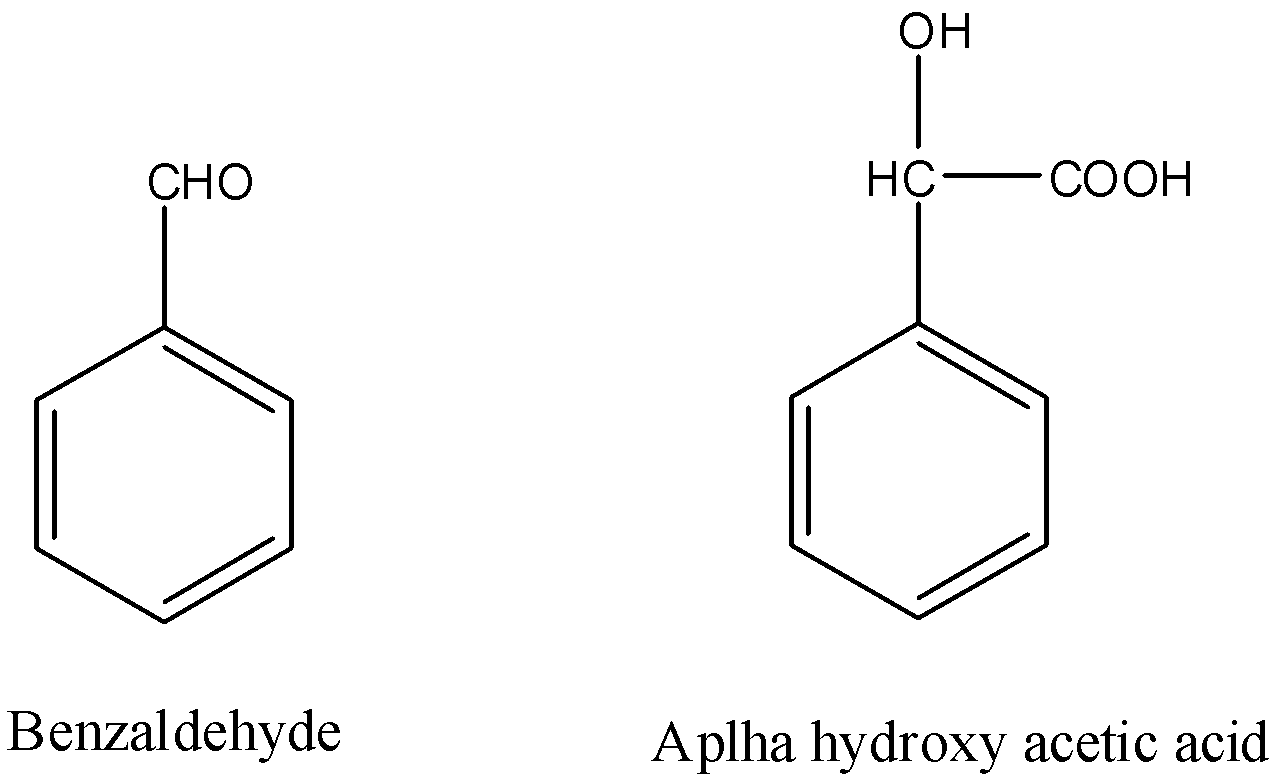
Benzaldehyde to \[\alpha \]-hydroxyphenylacetic acid.
Answer
615.6k+ views
Hint: The structures of Benzaldehyde and \[\alpha \]-hydroxyphenylacetic acid are as follows. So as we can see that one carbon has increased in the chain and that carbon is actually a carboxylic acid. We can use nucleophilic addition reactions to add a carbon atom in the aldehyde.
Complete step by step solution:
-The preparation of \[\alpha \]-hydroxyphenylacetic acid from benzaldehyde contains two steps.
Step-1:
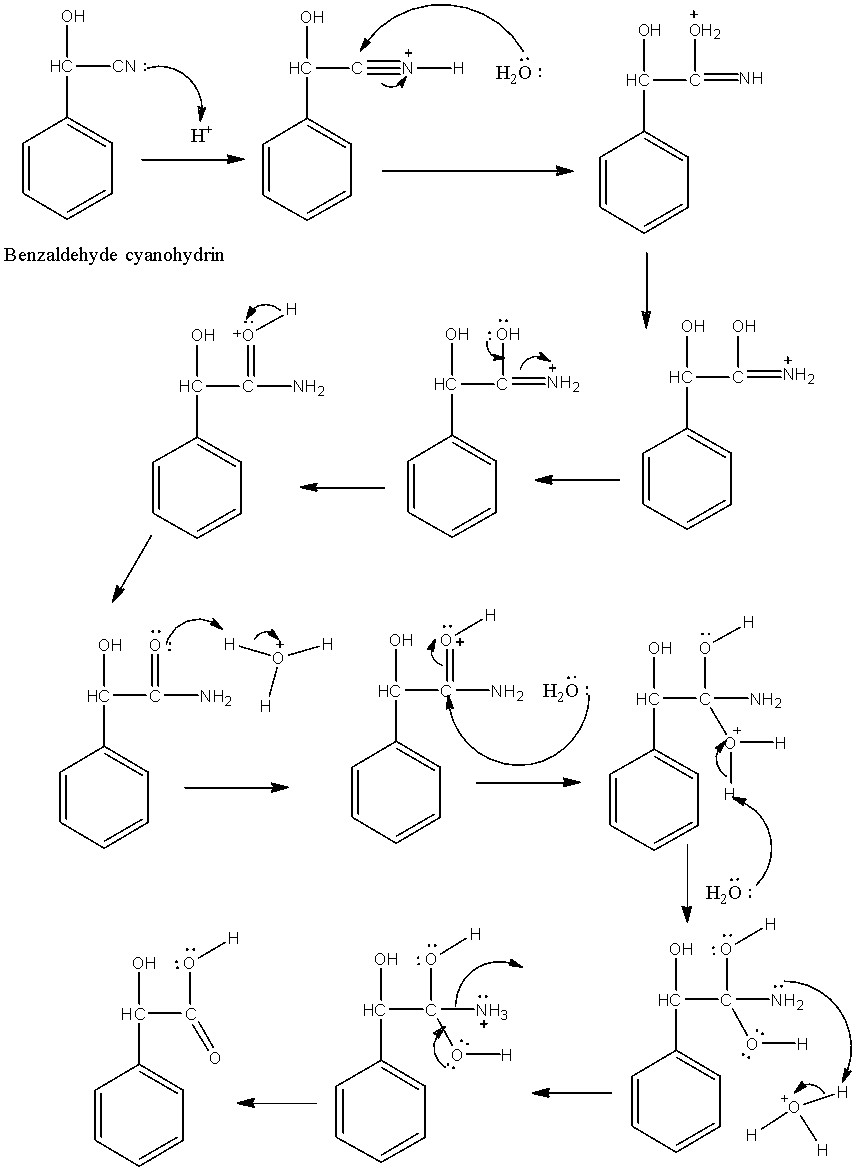
-Benzaldehyde undergoes nucleophilic addition reaction with sodium cyanide (NaCN) in the presence of hydrochloric acid and gives benzaldehyde cyanohydrin as the product at pH 9-10. Conversion of benzaldehyde to benzaldehyde cyanohydrin is as follows.
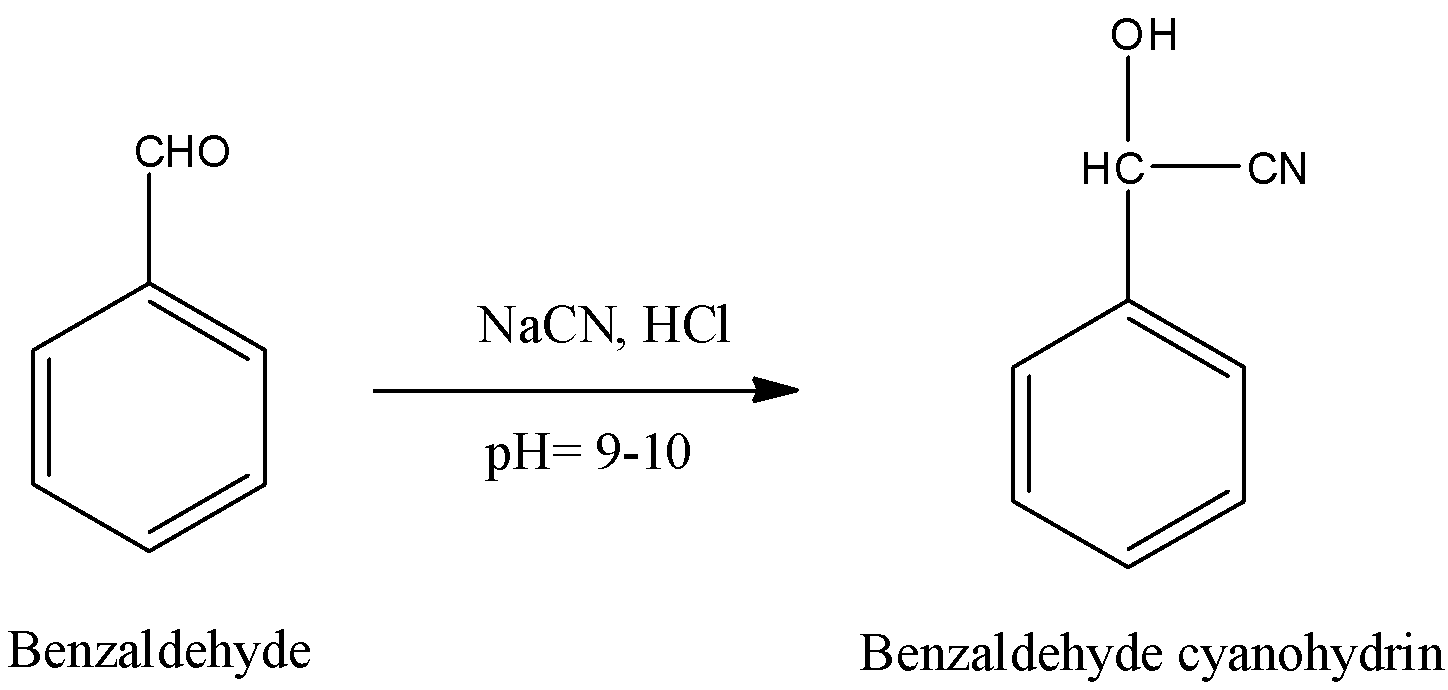
-In the above reaction Benzaldehyde undergoes bi molecular nucleophilic addition reaction with sodium cyanide to form benzaldehyde cyanohydrin as a product in step-1.
-The mechanism of nucleophilic addition reaction is as follows.
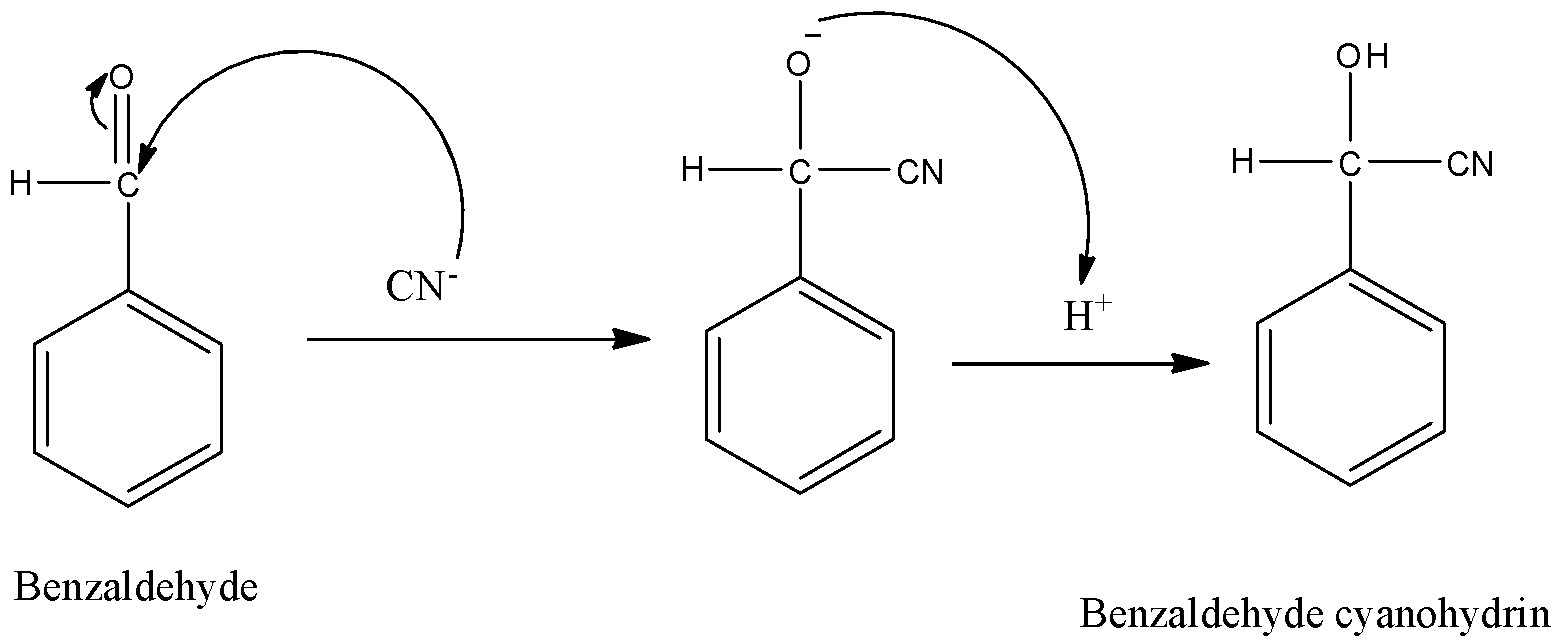
-Benzaldehyde cyanohydrin is also called as mandelonitrile.
Step-2:
-The formed benzaldehyde cyanohydrin undergoes hydrolysis in presence of acid and gives \[\alpha \]-hydroxyphenylacetic acid. Conversion of benzaldehyde cyanohydrin to \[\alpha \]-hydroxyphenylacetic acid is as follows.
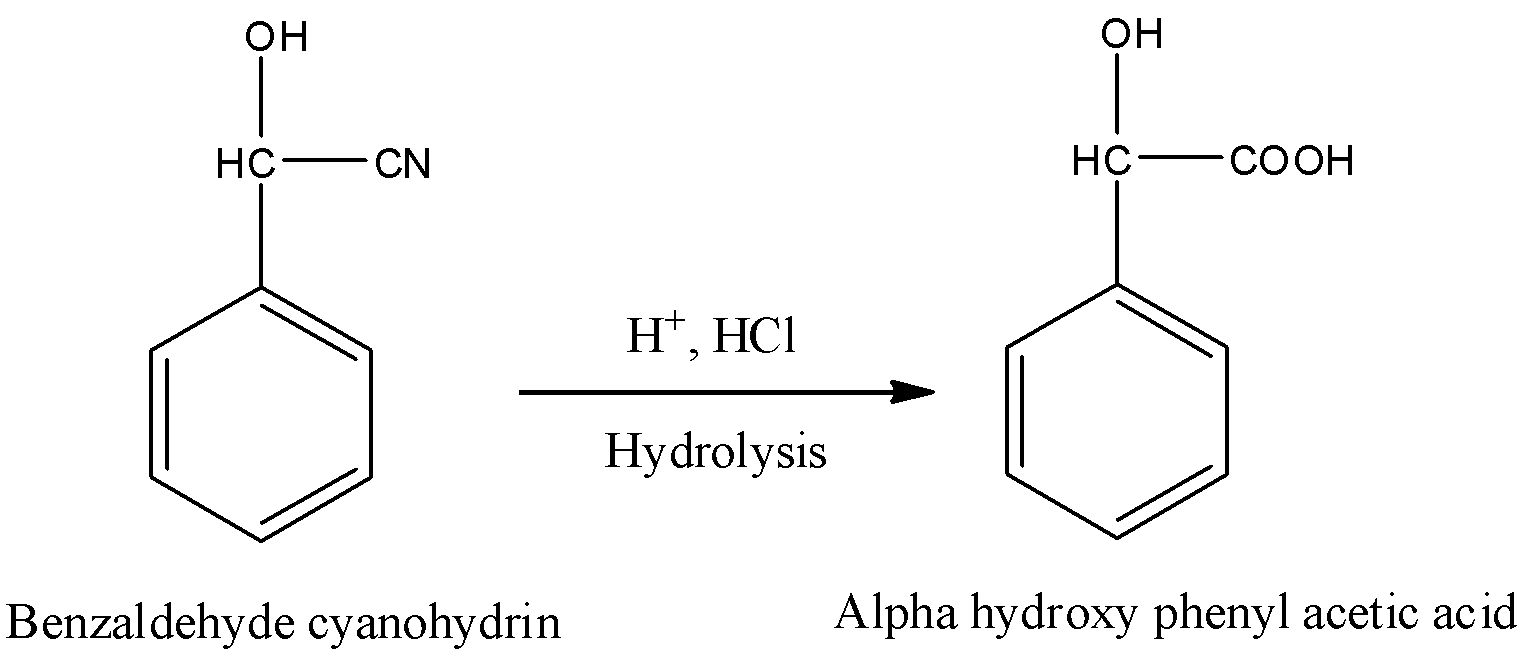
-In the above reaction cyanide undergoes hydrolysis in presence of acid and converts in to carboxylic acid in step-2.
-The mechanism of hydrolysis of cyanide is as follows.
Additional information:
-\[\alpha \]-hydroxyphenylacetic acid is also called as Mandelic acid and it is white crystalline solid.
-Mandelic acid is partially soluble in water due to the presence of carboxylic acid (-COOH) and hydroxyl (-OH) functional groups.
-Mandelic acid is completely soluble in organic solvents.
-Mandelic acid is used as a precursor in the preparation of drugs.
-Mandelic acid is used to treat urinary tract infections.
-Mandelic acid acts as an antibacterial agent.
Note: IUPAC name of\[\alpha \]-hydroxyphenylacetic acid is 2-hydroxy-2phenylacetic acid. \[\alpha \]-hydroxyphenylacetic acid has one chiral center in its structure. Generally \[\alpha \]-hydroxy acids (AHA) are used in cosmetics. Mandelic acid has two enantiomers called D- and L-Mandelic acid.

Complete step by step solution:
-The preparation of \[\alpha \]-hydroxyphenylacetic acid from benzaldehyde contains two steps.
Step-1:
-Benzaldehyde undergoes nucleophilic addition reaction with sodium cyanide (NaCN) in the presence of hydrochloric acid and gives benzaldehyde cyanohydrin as the product at pH 9-10. Conversion of benzaldehyde to benzaldehyde cyanohydrin is as follows.

-In the above reaction Benzaldehyde undergoes bi molecular nucleophilic addition reaction with sodium cyanide to form benzaldehyde cyanohydrin as a product in step-1.
-The mechanism of nucleophilic addition reaction is as follows.

-Benzaldehyde cyanohydrin is also called as mandelonitrile.
Step-2:
-The formed benzaldehyde cyanohydrin undergoes hydrolysis in presence of acid and gives \[\alpha \]-hydroxyphenylacetic acid. Conversion of benzaldehyde cyanohydrin to \[\alpha \]-hydroxyphenylacetic acid is as follows.

-In the above reaction cyanide undergoes hydrolysis in presence of acid and converts in to carboxylic acid in step-2.
-The mechanism of hydrolysis of cyanide is as follows.

Additional information:
-\[\alpha \]-hydroxyphenylacetic acid is also called as Mandelic acid and it is white crystalline solid.
-Mandelic acid is partially soluble in water due to the presence of carboxylic acid (-COOH) and hydroxyl (-OH) functional groups.
-Mandelic acid is completely soluble in organic solvents.
-Mandelic acid is used as a precursor in the preparation of drugs.
-Mandelic acid is used to treat urinary tract infections.
-Mandelic acid acts as an antibacterial agent.
Note: IUPAC name of\[\alpha \]-hydroxyphenylacetic acid is 2-hydroxy-2phenylacetic acid. \[\alpha \]-hydroxyphenylacetic acid has one chiral center in its structure. Generally \[\alpha \]-hydroxy acids (AHA) are used in cosmetics. Mandelic acid has two enantiomers called D- and L-Mandelic acid.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




