Aspartame, an artificial sweetener is a peptide and has the following structure. Which of the following is correct about the molecule?
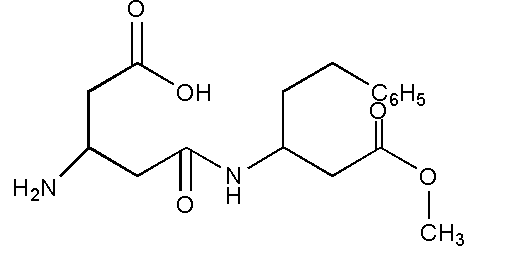
This question has multiple correct options.
A. It has four functional groups.
B. It has three functional groups
C. On hydrolysis it produces only one amino acid.
D. On hydrolysis it produces a mixture of amino acids.

Answer
588.9k+ views
Hint: Aspartame is used as a substitute of sugar as an artificial sweetener in foods and beverages. The aspartame contains two amino acids which are joined together by a peptide bond.
Complete step by step answer:
Aspartame is an artificial sweetener which is a non-saccharides which is 200 times sweeter than the sucrose. It is commonly used as a sugar substitute added in food and beverages. Aspartame is a methyl ester of aspartic acid. Aspartame is a formed in 1965 and approved for use in food products by FDA in 1981.
The structure of aspartame is shown below.
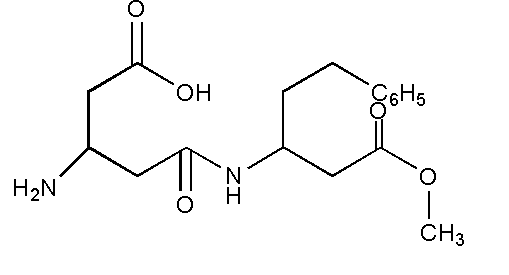
In the structure, it can be seen that the aspartame contains a total four functional groups. The functional groups are defined as those atoms or groups present inside the molecule which have their own characteristics property.
The functional groups present are amine \[ - N{H_2}\], carboxylic acid –COOH, ester \[ - COOR\], amide \[ - CONR\]
Aspartame on hydrolysis gives methanol, phenylalanine and aspartic acid as a product where phenylalanine and aspartic acid are two amino acids. Thus, on hydrolysis aspartame produces a mixture of two amino acids.
So, the correct answer is Option A,D.
Note: Aspartame due to its property of sweetener provides 4 kcal of energy per gram. The quantity of aspartame required to produce a sweet taste is small enough that its caloric value is negligible.
Complete step by step answer:
Aspartame is an artificial sweetener which is a non-saccharides which is 200 times sweeter than the sucrose. It is commonly used as a sugar substitute added in food and beverages. Aspartame is a methyl ester of aspartic acid. Aspartame is a formed in 1965 and approved for use in food products by FDA in 1981.
The structure of aspartame is shown below.

In the structure, it can be seen that the aspartame contains a total four functional groups. The functional groups are defined as those atoms or groups present inside the molecule which have their own characteristics property.
The functional groups present are amine \[ - N{H_2}\], carboxylic acid –COOH, ester \[ - COOR\], amide \[ - CONR\]
Aspartame on hydrolysis gives methanol, phenylalanine and aspartic acid as a product where phenylalanine and aspartic acid are two amino acids. Thus, on hydrolysis aspartame produces a mixture of two amino acids.
So, the correct answer is Option A,D.
Note: Aspartame due to its property of sweetener provides 4 kcal of energy per gram. The quantity of aspartame required to produce a sweet taste is small enough that its caloric value is negligible.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




