What are the products formed when $NaOH$ reacts with excess $S$ ?
(A) $N{a_2}{S_5},N{a_2}S{O_4}$
(B) $N{a_2}S,N{a_2}{S_2}{O_3}$
(C) $N{a_2}{S_5},N{a_2}{S_2}{O_3}$
(D) $N{a_2}{S_2}{O_3}$
Answer
596.7k+ views
Hint:We know that sodium hydroxide when boiled with an excess of sulfur, a disproportionate reaction occurs. In a disproportionate reaction, the same element in a compound is getting oxidized and reduced simultaneously. Here $S$ is undergoing a disproportionation reaction.
Complete answer:Sodium Hydroxide is an inorganic compound. It is also known as caustic soda. It is ionic and is a white crystalline solid. It has a formula $NaOH$ with cation $N{a^ + }$ and anion $O{H^ - }$ . Sulfur is a group $16$ element in the periodic table. It is non-metallic in nature and has a symbol $S$ . Sulfur is bright yellow, solid at room temperature. When sodium hydroxide is boiled with an excess of sulfur, the following reaction occurs:

It is a disproportionation reaction. A disproportionation reaction is a redox reaction in which the same element in a compound is getting oxidized and reduced simultaneously. So here sulfur $\left( S \right)$ is undergoing a disproportionation reaction. Its oxidation state changes from zero to $ - 2$ in sodium sulfide to $ + 4$ in sodium thiosulfate.
$N{a_2}S$ is sodium sulfide. It is a colorless solid and water-soluble. And it gives a strong alkaline solution when dissolved in water.
$N{a_2}{S_2}{O_3}$ is called sodium thiosulfate. It is an inorganic compound that is used as an antidote in cyanide poisoning and also an antifungal drug.
Thus, the correct option is $A$.
Note:It is important to write the correct stoichiometric coefficients in the equation. Different reactions occur if we change the stoichiometric coefficients. Look at the following equations:
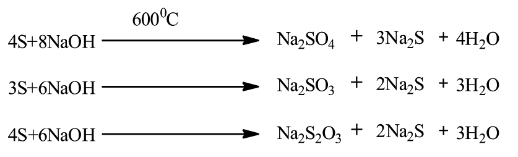
So if we compare these three equations, we can see that different products are formed if we change the stoichiometric coefficients.
Complete answer:Sodium Hydroxide is an inorganic compound. It is also known as caustic soda. It is ionic and is a white crystalline solid. It has a formula $NaOH$ with cation $N{a^ + }$ and anion $O{H^ - }$ . Sulfur is a group $16$ element in the periodic table. It is non-metallic in nature and has a symbol $S$ . Sulfur is bright yellow, solid at room temperature. When sodium hydroxide is boiled with an excess of sulfur, the following reaction occurs:

It is a disproportionation reaction. A disproportionation reaction is a redox reaction in which the same element in a compound is getting oxidized and reduced simultaneously. So here sulfur $\left( S \right)$ is undergoing a disproportionation reaction. Its oxidation state changes from zero to $ - 2$ in sodium sulfide to $ + 4$ in sodium thiosulfate.
$N{a_2}S$ is sodium sulfide. It is a colorless solid and water-soluble. And it gives a strong alkaline solution when dissolved in water.
$N{a_2}{S_2}{O_3}$ is called sodium thiosulfate. It is an inorganic compound that is used as an antidote in cyanide poisoning and also an antifungal drug.
Thus, the correct option is $A$.
Note:It is important to write the correct stoichiometric coefficients in the equation. Different reactions occur if we change the stoichiometric coefficients. Look at the following equations:

So if we compare these three equations, we can see that different products are formed if we change the stoichiometric coefficients.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




