How are the molecules arranged in the three states of matter?
Answer
585k+ views
Hint:The main three states of matter are solid, liquid, and gas. The matter is classified into these three states because of the nature of the arrangement of the particles and the intermolecular forces existing between the constituents of the matter.The strength of the intermolecular forces existing between the constituents of the matter decides the state of the matter.
Complete step-by-step answer:The three states the matters are solids, liquids, and gases.The diagrammatic representation of the solid particles is as follows:
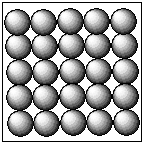
In the case of the solid, the constituent particles that are atoms, ions, molecules are held together by the strong intermolecular forces of the attraction. Hence, the particles of the solid substance are closely packed in a defined manner. This gives the shape of the particles to the solid substances.The diagrammatic representation of the liquid particles is as follows:
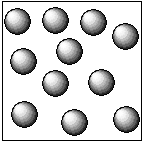
In the case of the liquid, the constituent particles that are atoms, ions, and molecules are held together by the intermolecular forces of the attraction which are weak compared to existing in the solid particles. Hence, the particles of the liquid substance are not closely packed. Therefore, the liquid does not possess a definite shape and volume. They occupy the shape of the container in which it is placed.The diagrammatic representation of the gas particles is as follows:
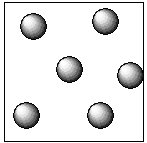
In the case of gases, the constituent particles that are atoms, ions, and molecules are held together by the weak intermolecular forces of the attraction which are weaker compared to existing in the liquid particles. Hence, the particles of the gaseous substance are not closely packed. Therefore, gases do not possess a definite shape and volume.
Note:The forces of the attraction existing between the particles of the same substance are known as intermolecular forces of the attraction.
The stronger the intermolecular forces more tightly will be the particles are arranged in the matter.
The different types of intermolecular forces include the different types of chemical bonds like hydrogen bond, covalent bond, ionic bond, metallic bond, the coordinate bond also weak forces like van der Waals forces, dipole-dipole interaction, London forces, etc. All these are leads to give specific shape and state to the matter.
Complete step-by-step answer:The three states the matters are solids, liquids, and gases.The diagrammatic representation of the solid particles is as follows:

In the case of the solid, the constituent particles that are atoms, ions, molecules are held together by the strong intermolecular forces of the attraction. Hence, the particles of the solid substance are closely packed in a defined manner. This gives the shape of the particles to the solid substances.The diagrammatic representation of the liquid particles is as follows:

In the case of the liquid, the constituent particles that are atoms, ions, and molecules are held together by the intermolecular forces of the attraction which are weak compared to existing in the solid particles. Hence, the particles of the liquid substance are not closely packed. Therefore, the liquid does not possess a definite shape and volume. They occupy the shape of the container in which it is placed.The diagrammatic representation of the gas particles is as follows:

In the case of gases, the constituent particles that are atoms, ions, and molecules are held together by the weak intermolecular forces of the attraction which are weaker compared to existing in the liquid particles. Hence, the particles of the gaseous substance are not closely packed. Therefore, gases do not possess a definite shape and volume.
Note:The forces of the attraction existing between the particles of the same substance are known as intermolecular forces of the attraction.
The stronger the intermolecular forces more tightly will be the particles are arranged in the matter.
The different types of intermolecular forces include the different types of chemical bonds like hydrogen bond, covalent bond, ionic bond, metallic bond, the coordinate bond also weak forces like van der Waals forces, dipole-dipole interaction, London forces, etc. All these are leads to give specific shape and state to the matter.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Convert 200 Million dollars in rupees class 7 maths CBSE

Full Form of IASDMIPSIFSIRSPOLICE class 7 social science CBSE

100 million is equal to begingathered A 1 crore B -class-7-maths-CBSE

List of coprime numbers from 1 to 100 class 7 maths CBSE

The least number which is a perfect square and is divisible class 7 maths CBSE

There are three types of tickets economy business and class 7 maths CBSE





