What are free radicals? Draw the structure of various types of free radicals and arrange them in increasing order of their stability.
Answer
575.7k+ views
Hint: We have heard about free radicals several times. They have a major role in the atmospheric industry, combustion, polymerization, biochemistry etc. They are also useful in several biological processes. They have a role in biological metabolism.
Complete step by step answer:
As it is familiar that the species of molecules having an unpaired electron are termed as free radicals. They are represented as \[{{{X}}^ \bullet }\] and are highly reactive species. Photo irradiation is a common method to produce free radicals. Light or heat in the form of energy is needed for the production of free radicals. Free radicals are classified into $\sigma $ and $\pi $ radicals. The unpaired electron is in $\sigma $ orbitals in $\sigma $ radicals. E.g. phenyl radical, vinyl radical etc. While the unpaired electron is in $\pi $ orbitals in $\pi $ radicals. E.g. t-butyl radical. $\pi $ radicals are generally stabilized by hyperconjugation effect or resonance effect. While there is no such stabilizing effect in $\sigma $ radicals.
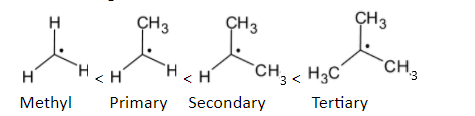
In the case of alkyl groups, when the number of alkyl groups on the carbon having free radicals is increased, the stability is increased.
The order of stability is given below:
Methyl radicalThe electron-deficient radical can be delocalized on the multiple bonds with carbon atoms. Stability is increased when a partially filled p orbital is participated in resonance with the adjacent p orbitals. The structure is given below:
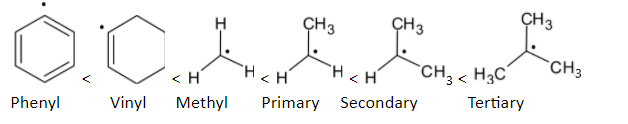
When we compare the stability of phenyl and vinyl radicals, the order is given below:
Note: The stability of free radicals depends upon three factors:
Increase in the number of alkyl groups on the carbon atom which has free radical.
Delocalization or resonance
Geometry of free radicals
Complete step by step answer:
As it is familiar that the species of molecules having an unpaired electron are termed as free radicals. They are represented as \[{{{X}}^ \bullet }\] and are highly reactive species. Photo irradiation is a common method to produce free radicals. Light or heat in the form of energy is needed for the production of free radicals. Free radicals are classified into $\sigma $ and $\pi $ radicals. The unpaired electron is in $\sigma $ orbitals in $\sigma $ radicals. E.g. phenyl radical, vinyl radical etc. While the unpaired electron is in $\pi $ orbitals in $\pi $ radicals. E.g. t-butyl radical. $\pi $ radicals are generally stabilized by hyperconjugation effect or resonance effect. While there is no such stabilizing effect in $\sigma $ radicals.
In the case of alkyl groups, when the number of alkyl groups on the carbon having free radicals is increased, the stability is increased.
The order of stability is given below:
Methyl radical

When we compare the stability of phenyl and vinyl radicals, the order is given below:

Note: The stability of free radicals depends upon three factors:
Increase in the number of alkyl groups on the carbon atom which has free radical.
Delocalization or resonance
Geometry of free radicals
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




