What are biodegradable and non-biodegradable detergents? Give one example of each.
Answer
621.9k+ views
Hint: The biodegradable, and non-biodegradable detergents can be defined in consideration with the various factors like structure, or it can be destroyed or not.
Complete step by step answer:
Now, first let us know about the detergents. As we know detergents are the cleansing agents as we use it to clean the clothes.Thus, we can specify the detergents like laundry detergents, or the dish detergents.Detergents are available in the powder form.
So, the detergents are classified into two types i.e. biodegradable, and non-biodegradable detergents.
-First, let us define the biodegradable detergents. These detergents are the straight hydrocarbon chain molecules.
From the biodegradable word we can say that these types of detergents are degraded; it means that they can be destroyed by the bacteria, or any other agents.
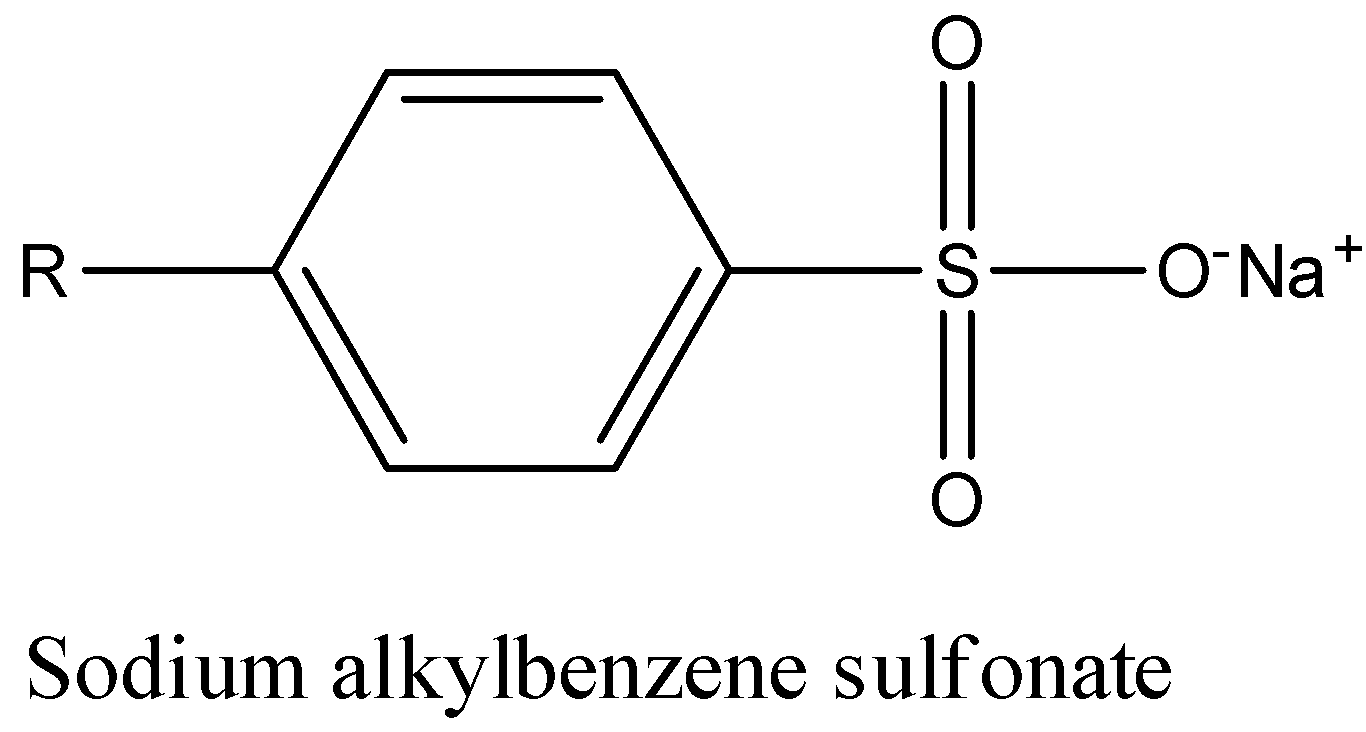
We have sodium alkyl benzene sulphonate as an example of biodegradable detergents.
The structure of the above is shown below-
-Second, we have non-biodegradable detergents. These detergents exist in branched hydrocarbon chains.
As we know that non-biodegradable relates to the non-destructive, and leads to the cause of pollution. So, non-biodegradable detergents cannot be destroyed by bacteria.
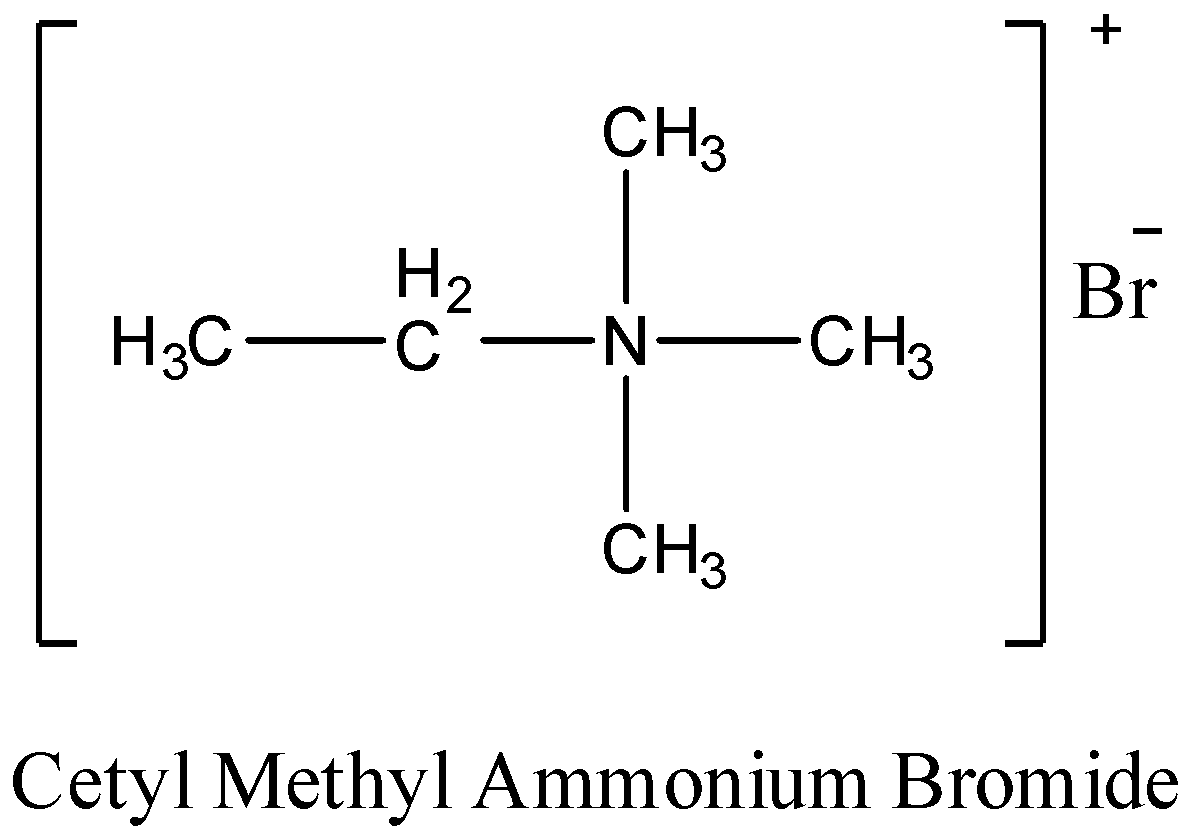
Cetyl methyl ammonium bromide as an example of non-biodegradable detergents. The structure of the above compound is shown below-
Note:
Synthetic detergents are further classified into three type on the basis of their charges
(i) Cationic detergents- These are basically quaternary ammonium salts of bromides, chlorides and acetates.
(ii) Anionic detergents- It is a long chain of sodium alcohol salts.
(iii) Non-ionic detergents.-These are high molecular mass esters with no ions present in the molecules.
Complete step by step answer:
Now, first let us know about the detergents. As we know detergents are the cleansing agents as we use it to clean the clothes.Thus, we can specify the detergents like laundry detergents, or the dish detergents.Detergents are available in the powder form.
So, the detergents are classified into two types i.e. biodegradable, and non-biodegradable detergents.
-First, let us define the biodegradable detergents. These detergents are the straight hydrocarbon chain molecules.
From the biodegradable word we can say that these types of detergents are degraded; it means that they can be destroyed by the bacteria, or any other agents.
We have sodium alkyl benzene sulphonate as an example of biodegradable detergents.
The structure of the above is shown below-

-Second, we have non-biodegradable detergents. These detergents exist in branched hydrocarbon chains.
As we know that non-biodegradable relates to the non-destructive, and leads to the cause of pollution. So, non-biodegradable detergents cannot be destroyed by bacteria.
Cetyl methyl ammonium bromide as an example of non-biodegradable detergents. The structure of the above compound is shown below-

Note:
Synthetic detergents are further classified into three type on the basis of their charges
(i) Cationic detergents- These are basically quaternary ammonium salts of bromides, chlorides and acetates.
(ii) Anionic detergents- It is a long chain of sodium alcohol salts.
(iii) Non-ionic detergents.-These are high molecular mass esters with no ions present in the molecules.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




