An excited $H{e^ + }$ ion emits two photons in succession, with wavelengths 108.5 nm and 30.4 nm, in making a transition to ground state. The quantum number n, corresponding to its initial excited state is (for photon of wavelength $\lambda $, energy $E = \dfrac{{1240}}{{\lambda ({\text{in nm}})}}eV$)
A. n = 5
B. n = 4
C. n = 6
D. n = 7
Answer
591.9k+ views
Hint: The total energy emitted by the two de-excitations is equal to the sum of the energy of the two de-excitations and the Bohr model gives expression for total energy emitted when a hydrogen-like atom undergoes de-excitation from a higher energy state to lower energy state. By equating this formula and the sum of the given energies, we can find out the required value of n for the excited state.
Formula used:
According to Bohr model, the expression for total energy emitted when a hydrogen-like atom undergoes de-excitation from a higher energy state to lower energy state is given by the following formula.
$E = 13.6{Z^2}\left[ {\dfrac{1}{{n_1^2}} - \dfrac{1}{{n_2^2}}} \right]eV$
The energy is given in terms of the wavelength of a light by the following expression.
$E = \dfrac{{hc}}{\lambda }$
Complete step by step answer:
We are given an excited \[H{e^ + }\] ion which is a hydrogen-like atom. It emits two photons in succession while making a transition to the ground state from the excited state. The two wavelengths emitted are given as
$
{\lambda _1} = 108.5nm \\
{\lambda _2} = 30.4nm \\
$
The following diagram shows the energy levels for the given atom where the de-excitation takes from an energy level n to the ground state n = 1
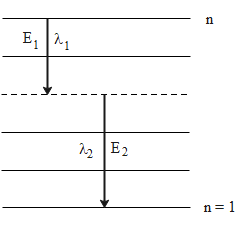
The energy emitted by a hydrogen-like atom in a single de-excitation from an excited state ${n_2}$ to a lower energy state ${n_1}$ is given by the following formula.
$E = 13.6{Z^2}\left[ {\dfrac{1}{{n_1^2}} - \dfrac{1}{{n_2^2}}} \right]eV$
Now for the given atom, Z = 2 and the total energy emitted by the two de-excitations is equal to the sum of the energy of the two de-excitations. Therefore, we have
$
E = {E_1} + {E_2} \\
\Rightarrow 13.6{Z^2}\left[ {\dfrac{1}{{{1^2}}} - \dfrac{1}{{{n^2}}}} \right] = \dfrac{{hc}}{{{\lambda _1}}} + \dfrac{{hc}}{{{\lambda _2}}} \\
\Rightarrow 13.6 \times {\left( 2 \right)^2}\left[ {\dfrac{1}{{{1^2}}} - \dfrac{1}{{{n^2}}}} \right] = \dfrac{{1240eV/nm}}{{108.5nm}} + \dfrac{{1240eV/nm}}{{30.4nm}} = 11.43 + 40.79 = 52.22 \\
\Rightarrow 1 - \dfrac{1}{{{n^2}}} = \dfrac{{52.22}}{{54.4}} \\
\Rightarrow \dfrac{1}{{{n^2}}} = 1 - \dfrac{{52.22}}{{54.4}} = \dfrac{{2.18}}{{54.4}} \\
\Rightarrow {n^2} = \dfrac{{54.4}}{{2.18}} = 25 \\
\therefore n = 5 \\
$
This is the required value of n for the excited state. Hence, the de-excitation takes from n = 5 state, i.e. the fourth excited state,
So, the correct answer is option A.
Note:
Hydrogen-like atoms are those which have the same electronic configuration as that of a neutral hydrogen atom. The neutral helium atom is not hydrogen-like but if we remove one electron from it then we get the same electronic configuration as hydrogen. Similarly, other hydrogen-like atoms are $L{i^{2 + }}$, $B{e^{3 + }}$, etc.
Formula used:
According to Bohr model, the expression for total energy emitted when a hydrogen-like atom undergoes de-excitation from a higher energy state to lower energy state is given by the following formula.
$E = 13.6{Z^2}\left[ {\dfrac{1}{{n_1^2}} - \dfrac{1}{{n_2^2}}} \right]eV$
The energy is given in terms of the wavelength of a light by the following expression.
$E = \dfrac{{hc}}{\lambda }$
Complete step by step answer:
We are given an excited \[H{e^ + }\] ion which is a hydrogen-like atom. It emits two photons in succession while making a transition to the ground state from the excited state. The two wavelengths emitted are given as
$
{\lambda _1} = 108.5nm \\
{\lambda _2} = 30.4nm \\
$
The following diagram shows the energy levels for the given atom where the de-excitation takes from an energy level n to the ground state n = 1

The energy emitted by a hydrogen-like atom in a single de-excitation from an excited state ${n_2}$ to a lower energy state ${n_1}$ is given by the following formula.
$E = 13.6{Z^2}\left[ {\dfrac{1}{{n_1^2}} - \dfrac{1}{{n_2^2}}} \right]eV$
Now for the given atom, Z = 2 and the total energy emitted by the two de-excitations is equal to the sum of the energy of the two de-excitations. Therefore, we have
$
E = {E_1} + {E_2} \\
\Rightarrow 13.6{Z^2}\left[ {\dfrac{1}{{{1^2}}} - \dfrac{1}{{{n^2}}}} \right] = \dfrac{{hc}}{{{\lambda _1}}} + \dfrac{{hc}}{{{\lambda _2}}} \\
\Rightarrow 13.6 \times {\left( 2 \right)^2}\left[ {\dfrac{1}{{{1^2}}} - \dfrac{1}{{{n^2}}}} \right] = \dfrac{{1240eV/nm}}{{108.5nm}} + \dfrac{{1240eV/nm}}{{30.4nm}} = 11.43 + 40.79 = 52.22 \\
\Rightarrow 1 - \dfrac{1}{{{n^2}}} = \dfrac{{52.22}}{{54.4}} \\
\Rightarrow \dfrac{1}{{{n^2}}} = 1 - \dfrac{{52.22}}{{54.4}} = \dfrac{{2.18}}{{54.4}} \\
\Rightarrow {n^2} = \dfrac{{54.4}}{{2.18}} = 25 \\
\therefore n = 5 \\
$
This is the required value of n for the excited state. Hence, the de-excitation takes from n = 5 state, i.e. the fourth excited state,
So, the correct answer is option A.
Note:
Hydrogen-like atoms are those which have the same electronic configuration as that of a neutral hydrogen atom. The neutral helium atom is not hydrogen-like but if we remove one electron from it then we get the same electronic configuration as hydrogen. Similarly, other hydrogen-like atoms are $L{i^{2 + }}$, $B{e^{3 + }}$, etc.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




